Thank you for attending PNHP’s 2022 Annual Meeting, Leadership Training, and/or SNaHP Summit. Please complete the relevant evaluations below, based on which sessions you attended.
If you have additional feedback, please email organizer@pnhp.org.
Thank you for attending PNHP’s 2022 Annual Meeting, Leadership Training, and/or SNaHP Summit. Please complete the relevant evaluations below, based on which sessions you attended.
If you have additional feedback, please email organizer@pnhp.org.
Click the links below to jump to different sections of the newsletter. To view a PDF version of the shorter print edition of the newsletter, click HERE.
If you wish to support PNHP’s outreach and education efforts with a financial contribution, click HERE.
If you have feedback about the newsletter, email info@pnhp.org.
After two years of meeting virtually, PNHP members and allies will once again gather in-person for our Annual Meeting and Leadership Training. This year’s conference, themed “Brick by Brick: Building the Movement for Medicare for All,” will take place Nov. 4-5 at the Boston Park Plaza.
Our keynote speakers for Boston will address some of the most far-reaching and pressing issues facing the Medicare-for-All movement. Linda Villarosa, contributor to The New York Times’ 1619 Project and author of the just-published Under the Skin: The Hidden Toll of Racism on American Lives and the Health of Our Nation will discuss the necessity of confronting systemic racism as part of any effort to achieve health justice, and Donald Cohen, co-author of The Privatization of Everything, will discuss the profiteering that threatens Medicare and Medicaid alongside every other public good in the United States.

PNHP is also thrilled to be hosting the Students for a National Health Program (SNaHP) Summit on Nov. 4, immediately preceding this year’s Leadership Training. Typically held in the spring, the SNaHP Summit is a chance for medical and health professional students to meet like-minded colleagues, strategize for the year ahead, and organize around tangible goals that advance the single-payer movement, both inside and outside of their institutions.
It’s been three long years since PNHP members have been able to gather in-person for an Annual Meeting. In that time, the work of our movement has become more urgent than ever; please join us as we engage in a long overdue weekend of learning, connecting, and strategizing for our shared goal: single-payer Medicare for All.
Online pre-registration is open through Sunday, October 30 at pnhp.org/meeting.
PNHP’s Board of Directors and medical experts have established Covid safety protocols for the Annual Meeting that include a surgical facemask requirement while not actively eating or drinking; proof of vaccine within the last 5 months (physical card, photo, or electronic record is fine); and proof of a negative Covid test (time-stamped photo of a rapid antigen test is fine) taken within 24 hours of the Summit.
In addition, while the conference includes meals, times designated for food service will not contain programming so that participants can eat away from the main conference room should they choose.


By Suzanne Gordon
It’s no surprise to PNHP members that the same vested interests opposed to Medicare for All want to undermine the Veterans Health Administration (VHA), our largest public health care system.
Run by the Department of Veterans Affairs (VA), the VHA is our best working model of socialized medicine. VHA care-givers are not a collection of physician practices or specialty services reimbursed by private insurers, Medicare, or Medicaid on a “fee for service” basis. All VHA doctors, nurses, therapists, and other personnel are salaried, like members of the UK National Health Service.
VHA staff provide high-quality care to nine million patients that is better coordinated and more cost effective than in the private sector. The VHA plays a major “teaching hospital” role in training thousands of new doctors, nurses, and other health care professionals. It doesn’t compete with other hospital chains by spending huge sums on advertising and marketing.
In 2018, corporate Democrats and conservative Republicans took a sledge hammer to the VHA when they passed the VA MISSION Act. As implemented by Donald Trump and now Joe Biden, this legislation has siphoned billions of dollars away from the VHA’s direct care budget and steered that money toward private doctors and for-profit hospitals often less well prepared to treat veterans.
The VHA has been partially converted into a Medicare-style payer of bills submitted by other health care providers. The powerful private interests that have acquired this new $30 billion a year federal revenue stream want to preserve and expand it—just like operators of Medicare Advantage plans and newly minted ACO REACH entities hope to profit from further diversion of seniors from traditional Medicare.
The community-labor campaign to save the VHA from further privatization and the PNHP-led resistance to Medicare profiteering relies on many of the same allies and faces common adversaries. They are parallel struggles in the same fight to build on what’s best in American health care—and we’ll all have a better chance of winning if we work more closely together.
Healthcare journalist Suzanne Gordon is a longtime PNHP supporter and co-founder of the Veterans Healthcare Policy Institute. She will lead a workshop on protecting the VHA from profteers at the PNHP Annual Meeting on Nov. 5. Her new book is called “Our Veterans: Winners, Losers, Friends and Enemies on the New Terrain of Veterans Affairs” from Duke University Press.
When the U.S. Supreme Court issued its ruling in Dobbs v. Jackson Women’s Health Organization earlier this summer, overturning nearly 50 years of federal abortion protections, it prompted a wave of shock, anger, grief and, ultimately, organizing among Americans who care deeply about reproductive justice.
Against this backdrop, PNHP board advisers Drs. Diljeet Singh and Ashley Duhon, along with PNHP national board member Dr. Judith Albert, are planning a workshop for the Nov. 5 Annual Meeting in Boston. Their goal is to brainstorm appropriate responses to this severe curtailing of abortion access, seek ways to support patients and frontline providers, and amplify the work of abortion rights activists both inside and outside of the medical profession. Focusing intently on reproductive justice is both crucial and long overdue.
“We do not always focus on how equity is one of the important principles of single payer,” says Dr. Singh. “Now we have no choice but to talk about it and to rally around it.”
To read PNHP’s statement in response to the Dobbs decision, visit pnhp.org/AbortionStatement. To connect with fellow reproductive justice activists within PNHP, contact Dr. Singh at diljeetksingh@gmail.com.
At last year’s virtual Annual Meeting, PNHP launched a series of Member Interest Groups (MIGs) based on medical specialties, lived experiences, and areas of interest. One of these groups, our Pediatrics MIG, is planning to ramp up their activism with an in-person meeting in Boston on Nov. 5. At the top of their agenda will be organizing within the American Academy of Pediatrics as part of PNHP’s ongoing Medical Society Resolutions campaign.
“There are a lot of pediatricians within PNHP, but the Academy has been resistant to anything on single payer,” says Dr. Eve Shapiro, a PNHP national board member who is active within the MIG. Dr. Shapiro envisions brainstorming with her colleagues in Boston to better understand roadblocks within the Academy, organize a sign-on letter for pediatricians and, ultimately, win passage of a single-payer resolution.
“Seeing large medical organizations saying Medicare for All is a good idea is powerful,” she says. “It can move the issue forward.”
To get involved with the Pediatrics MIG, contact Dr. Shapiro at evecshapiro@gmail.com. For questions about other MIGs, contact Kaytlin Gilbert at kaytlin@pnhp.org. To learn more about organizing within medical societies, contact Lori Clark at lori@pnhp.org.
 Previous Experience: At the Jane Addams Senior Caucus, I built a powerful base of leaders who were bonded not by candidates or party, but by a vision for a better future.
Previous Experience: At the Jane Addams Senior Caucus, I built a powerful base of leaders who were bonded not by candidates or party, but by a vision for a better future.
What drew you to PNHP? I wanted to join an organization that was interested in investing in the development of its members, building power, and developing a roadmap to take back control of our health care system from big corporations and the insurance industry.
What are you looking forward to working on over the next 12 months? I am looking forward to working together to create a new organizing model that grows PNHP’s collective power, and helping to develop a strategic roadmap that advances the single-payer movement.
What’s a fun fact about yourself? I lived in an apartment for over a year before I knew that the oven did not work.
Connect with Lori at lori@pnhp.org.
 Previous Experience: I previously worked in nuclear and cyber policy research at the Carnegie Endowment for International Peace in Washington, D.C.
Previous Experience: I previously worked in nuclear and cyber policy research at the Carnegie Endowment for International Peace in Washington, D.C.
What drew you to PNHP? As a progressive, I believe that implementing Medicare for All is the best way to fix our country’s deeply flawed health care system. I myself come from a family of doctors, so I’ve always had an appreciation for their leadership and impact in the communities they serve, and I know that if they advocate for single payer, people will listen.
What are you looking forward to working on over the next 12 months? I’m most looking forward to working on our campaign to stop the privatization of Medicare, as well as planning for our annual meeting in November!
What’s a fun fact about yourself? Every year, I go to a documentary film festival in Columbia, Mo. known as True/False.
Connect with Gaurav at gaurav@pnhp.org.
98 million skipped care or cut back on basic needs: Health care costs pushed 38% of American adults to delay or skip health care, cut back on driving, utilities or food, or borrow money to pay for medical bills in the first half of 2022, including 26% who only delayed or avoided care. Lower-income households were hit hardest, with 62% making cutbacks. But high income households were not immune, with 19% of households making at least $180,000 cutting back. “Estimated 98 Million Americans Skipped Treatments, Cut Back on Food, Gas or Utilities to Pay for Healthcare,” West Health-Gallup, 8/4/2022
Americans with serious illness unable to get care: Among households where a member has been seriously ill in the past year, 19% report they delayed care or were unable to get care when they needed it, including 24% of Black households. Lack of health insurance was not the primary factor: more than 80% reported having health insurance. Black respondents reported greater rates of poor treatment, with 15% saying they were disrespected, turned away, unfairly treated, or received poor treatment because of their race or ethnicity, compared with 3% of white respondents. “Personal Experiences of U.S. Racial/Ethnic Minorities in Today’s Difficult Times,” NPR-Robert Wood Johnson Foundation-Harvard T.H. Chan School of Public Health, August 2022
Older adults skip basics to pay for health care: Americans aged 50 and older report forgoing needed treatments because of the cost of care, including 26% of adults age 50 to 64 and 12% of adults 65 and older. Similarly, 18% of people 50 to 64 and 11% of people 65 and older report skipping medication to save money. Large majorities of older adults say health care costs are a major or minor burden, including 72% of people age 50 to 64 (24% major burden, 48% minor burden) and 66% of people 65 and older (15% major burden, 51% minor burden). Willcoxon, “Older Adults Sacrificing Basic Needs Due to Healthcare Costs,” Gallup, 6/15/2022
Insurance is third greatest living expense: Health insurance premiums account for 10.69% of an average U.S. salary, costing $6,487.20. This was the highest living expense after rent and childcare. Health insurance costs relative to salary vary by geography: in four states, insurance costs average at 15% of salary or more, including West Virginia at 20.9%. “Salary vs Health Insurance,” NiceTX, July 2022
Majority of U.S. adults had medical debt in past 5 years: A Kaiser Family Foundation survey finds 41% of adults currently have debt due to medical or dental bills, and an additional 16% have had medical or dental debt in the past five years that has since been paid off. Among subgroups reporting current medical debt are 56% of Black adults, 50% of Hispanic adults, 26% of households earning $90,000 or more, and 22% of adults age 65 and older. Medical debt is less common in states that expanded Medicaid (39%) than in states that did not (47%). Medical debt also forces households to change their behaviors, with 63% of adults with medical debt saying they cut back on spending for food or other basic household items, 40% who took an extra job or worked more hours, and 28% who delayed a home purchase or education for a member of their household. Lopes et al., “Health Care Debt in the U.S.: The Broad Consequences of Medical and Dental Bills,” Kaiser Family Foundation, 6/16/2022
Even the best U.S. states are outpaced by peer countries: A Commonwealth Fund analysis finds that Americans – regardless where they live – have lower life expectancy and greater incidence of avoidable mortality than other middle- and high-income countries. The U.S. ranked 31st among 38 peer countries on life expectancy at birth (78.8 years in 2019) and avoidable deaths before age 75 (272 per 100,000). State level analysis shows that even the states with the best outcomes are below average compared with peer countries, and the states with the lowest life expectancies and highest rates of avoidable deaths have worse outcomes than the worst-ranked peer countries. Radley et al., “Americans, No Matter the State They Live in, Die Younger Than People in Many Other Countries,” To the Point (blog), Commonwealth Fund, 8/11/2022
U.S. spends heavily on cancer care, gets middling outcomes: Among 22 high-income countries, the U.S. has the highest spending on cancer care – over $200 billion in 2020, or $584 per person – yet overall cancer mortality is only slightly better than average. Median per capita spending among the 22 countries was $296. After adjustments for smoking rates, U.S. cancer mortality was higher than nine other countries, and researchers found no association between cancer care expenditures and cancer mortality. Chow et al., “Comparison of Cancer-Related Spending and Mortality Rates in the US vs 21 High-Income Countries,” JAMA Health Forum, 5/27/22
U.S. men report poorer health, more cost-related access problems: A survey of men in 11 high-income countries shows U.S. men have the highest rates of avoidable deaths (337 per 100,000 vs. 156-233), multiple chronic conditions (29% vs. 17-25%), and hypertension (37% vs. 20-28%).The U.S. was tied with Switzerland for having the highest out-of-pocket health spending, with 33% reporting having out-of-pocket costs of $2,000 or more, and 37% reporting cost-related access problems, compared with 7% to 25% in other countries. U.S. men were second behind Australia for having mental health needs, and second behind Sweden for not having a regular doctor or place of care. Only 37% of U.S. men rate their country’s health care system as “good” or “very good” compared with 60% to 88% of men in other countries. Gunja et al., “Are Financial Barriers Affecting the Health Care Habits of American Men?” Commonwealth Fund, 7/14/2022
Diabetics face catastrophic insulin costs: Among the 7 million Americans who use daily insulin, 14.1% spent more than 40% of their post-subsistence income (after food and housing) on insulin, considered a “catastrophic” level. Patients with private insurance or no insurance paid the most out of pocket, while Medicaid beneficiaries were 61% less likely to have catastrophic spending. Bakkila et al., “Catastrophic Spending on Insulin in the United States, 2017-18,” Health Affairs, July 2022
Majority of hepatitis C patients don’t get curative treatment: Despite having a treatment that cures more than 95% of patients with hepatitis C, less than one-third of infected people with insurance get the treatment. The lowest rates of treatment were among adults aged 18-29 and Medicaid recipients. Among Medicaid patients, treatment rates were lower for Black patients and in programs with treatment restrictions. Coverage restrictions can include preauthorization requirements, clinical or social eligibility restrictions, or medical specialist prescribing. Prevalence of treatment within 360 days of a positive test were 23% for patients with Medicaid, 28% with Medicare, and 35% with private insurance. Thompson et al., “Vital Signs: Hepatitis C Treatment Among Insured Adults — United States, 2019-2020,” Centers for Disease Control and Prevention MMWR, 8/12/2022
Childbirth is costly despite insurance: Health care costs for women who give birth average $18,865 more than for women who do not give birth, including $2,854 more in out of pocket expenses. The analysis of women in large group health plans estimated the health costs associated with pregnancy, delivery, and postpartum care. Rae et al., “Health costs associated with pregnancy, childbirth, and postpartum care,” Peterson-Kaiser Family Foundation Health System Tracker, 7/13/2022
Physician face time increases, but disparities grow: Over the past 40 years, the amount of time patients spend annually with outpatient physicians increased to 60.4 minutes in 2018, up from 40 minutes in 1978, mainly due to an increase in average visit duration (15.4 minutes to 22.2 minutes). Time with primary care physicians fell, however, from 33.8 minutes to 30.4 minutes – owing to fewer primary care visits – while time spent with surgeons and medical specialists increased (12.1 to 12.6 minutes with surgeons, 15.4 to 17.4 minutes with medical specialists). While face time increased among all racial and ethnic groups, white patients continue to get more physician time than their Black and Hispanic counterparts. The white-Black gap increased from 13.1 to 22.9 minutes, while the white-Hispanic gap rose from 11.6 to 14.7 minutes. White patients spent significantly less time with specialists than their Black and Hispanic counterparts. Gaffney et al., “Trends and Disparities in the Distribution of Outpatient Physicians’ Annual Face Time with Patients, 1979-2018,” Journal of General Internal Medicine, 6/6/2022
Disparities, barriers persist despite insurance: Black patients enrolled in employer-sponsored commercial insurance are more likely to be burdened by chronic diseases, experience barriers to care, and have unmet social needs according to a study sponsored by Morgan Health, a JP Morgan Chase & Co. initiative. Black enrollees are 15.6 percentage points more likely than white enrollees to have uncontrolled high blood pressure and 5.3 percentage points more likely to have diabetes, after adjusting for age and sex. Black enrollees were 4.9 percentage points more likely to visit an emergency department and were 9.8 percentage points more likely to be food insecure. “Health Disparities in Employer-Sponsored Insurance,” Morgan Health and NORC, July 2022
More Black patients, less reimbursement: An analysis of Medicare data shows hospitals serving a disproportionate share of Black patients receive 21.6% lower payments for patient care per day than other hospitals. Hospitals serving Black patients averaged a loss of $17 per patient day versus an average profit of $126 at other hospitals. After adjusting for patient case mix and hospital characteristics, Black-serving hospitals still received $238 less in revenue per patient day than other hospitals, and $111 less in profits. Researchers estimate $14 billion would have been required to equalize reimbursement levels at Black-serving hospitals in 2018. Himmelstein et al., “Hospitals that Serve Many Black Patients Have Lower Revenues and Profits: Structural Racism in Hospital Financing,” Journal of General Internal Medicine, 8/5/2022
Medicaid utilization lower among Black patients: Medicaid spends an average of $317 less on Black enrollees than their white counterparts after adjusting for demographics, health status, and source of care. Black enrollees had fewer primary care encounters than white enrollees (19.3 fewer per 100 enrollees annually) but more emergency department visits (9.5 more per 100 enrollees). Among children, Black enrollees had 90.1 fewer primary care encounters per 100 enrollees. Wallace et al., “Disparities in Health Care Spending and Utilization Among Black and White Medicaid Enrollees,” JAMA Health Forum, 6/10/2022
Early Covid vaccine rollout plagued by disparities: U.S. health care facilities in counties with a high proportion of Black residents were less likely to receive Covid-19 vaccines in May 2021. Facilities in counties with at least 42.2% Black residents were less likely to administer Covid vaccines than facilities in counties with less than 12.5% Black residents. In urban areas, counties with large Black populations were 32% less likely to provide vaccinations than urban counties with low Black populations. Researchers also found facilities in rural counties and counties in the top quintile of Covid mortality were also less likely to administer Covid vaccines. In rural counties with large Hispanic populations, facilities were 26% less likely to administer vaccines than in rural counties with low Hispanic populations. Hernandez et al., “Disparities in distribution of COVID-19 vaccines across US counties: A geographic information system-based cross-sectional study,” PLOS Medicine, 7/28/2022
Disparities in monkeypox infections, vaccinations: Early data indicates racial disparities in monkeypox infections, unaddressed by vaccination. As of the end of July, 26% of known monkeypox cases were among Blacks and 32% were among Hispanics. Vaccinations have gone to whites, including 55% of vaccine recipients in Chicago and 63.5% in Washington, D.C. Black people are disproportionately diagnosed with monkeypox in North Carolina (70%) but only 22% of vaccine doses have gone to Black men, approximately mirroring the state’s population. In Georgia, 71% of cases have been among Black men, while only 44.5% of people vaccinated were Black. Johnson et al., “White People Get Bigger Share of Monkeypox Shots, Early Data Show,” Bloomberg, 8/11/2022; McFarling et al., “New data from several states show racial disparities in monkeypox infections,” STAT News, 8/11/2022; Pratt, “Racial disparities revealed in monkeypox vaccination data,” Atlanta Journal-Constitution, 8/18/2022
Disparities in drug overdose deaths: Deaths from unintentional or undetermined overdose increased by 44% among Black people compared with 22% for white people in 2020, Black incidence of death rose from 27 deaths per 100,000 people to 38.9 versus an increase from 25.2 to 30.7 for whites. Death rates also increased for American Indian or Alaska Native people by 39% (from 26.2 to 36.4) and 21% among Hispanics (17.3 to 21). Black youths age 15 to 24 saw an 86% increase in overdose deaths, the largest increase of any age or racial group. Black men 65 and older were nearly seven times as likely as white men to die from an overdose. At the same time, Black people were less than half as likely as white people to have received substance use treatment. Kariisa et al., “Vital Signs: Drug Overdose Deaths, by Selected Sociodemographic and Social Determinants of Health Characteristics — 25 States and the District of Columbia, 2019-2020,” Centers for Disease Control and Prevention MMWR, 7/19/2022
Single Payer could have saved thousands of lives: Single-payer health care could have prevented 338,594 Covid-19 deaths in the U.S. from the beginning of the public health emergency to mid-March 2022. Researchers estimate that if everyone in the country was provided with comprehensive care for free at the point of service, 131,438 people who died from Covid-19 could have been spared in 2020 alone, and roughly 80,000 people with other diseases could have been saved that year. More than 207,000 additional Covid-19 deaths could have been averted in 2021 and the first three months of this year. The U.S. also could have saved $105.6 billion in health care costs associated with hospitalizations from the disease—on top of the estimated $438 billion that could be saved in a non-pandemic year. Galvani et al., “Universal healthcare as pandemic preparedness: The lives and costs that could have been saved during the COVID-19 pandemic,” PNAS, 6/13/22
More non-Covid deaths in states with greater uninsurance: The White House Council of Economic Advisors found that states with high uninsurance rates had more non-Covid “excess deaths” during the first two years of the pandemic. They estimate that each 10 percentage point increase in a state’s uninsurance rate was associated with a 4.8 percentage point increase in excess deaths. “Excess Mortality during the Pandemic: The Role of Health Insurance,” White House Council of Economic Advisors, 7/12/2022
Uninsurance associated with late-stage cancer diagnosis, lower survival: A new study shows that people without insurance are significantly more likely to be diagnosed with late-stage cancers and face lower survival rates than their insured peers. The difference was particularly marked for six cancers – prostate, colon, non-Hodgkin lymphoma, oral cavity, liver, and esophagus – where uninsured individuals diagnosed with stage 1 disease fared worse than insured people diagnosed with stage 2 disease. The analysis suggested that people without health insurance were more likely to postpone doctor visits, resulting in a late-stage diagnosis, the researchers said. But people without health insurance coverage were also more likely to have worse short- and long-term survival rates after diagnosis. Uninsured individuals fared worse within each stage for all of the 19 cancers combined. Zhao et al., “Health insurance status and cancer stage at diagnosis and survival in the United States,” CA: A Cancer Journal for Clinicians, 7/13/2022
Suicide deaths rose less in states that expanded Medicaid: Suicide is the 10th leading cause of death in the U.S. and the second leading cause of death in people age 10 to 34. Suicides have been steadily increasing since 1999, with a mean increase of 1% per year from 1999 to 2006 and 2% per year from 2006 to 2018. However, researchers found that death by suicide increased less in states that expanded Medicaid coverage, suggesting the blunting of rising suicide rates among adults age 20 to 64 could be linked to better access to mental health care. Patel et al., “Association of State Medicaid Expansion Status with Rates of Suicide Among US Adults,” JAMA Network Open, 6/15/2022
Paid sick leave tied to fewer ER visits: From 2011 through 2019, in states that put paid sick leave policies in place, ED visits fell 5.6% — or about 23 fewer visits per 1,000 people per year. The biggest drops came from Medicaid patients, with big declines in visits that could have been handled in primary care: for adults, dental problems, mental health issues, and substance use disorder; and for kids, asthma. Ma et al., “State Mandatory Paid Sick Leave Associated with a Decline in Emergency Department Use in the US, 2011-19,” Health Affairs, August 2022
ACA insurers deny nearly one in five claims, but won’t tell patients why: ACA Marketplace insurers denied, on average, nearly one-fifth (18%) of in-network claims. And for 72% of denials, the explanation that insurers offered was “all other reasons.” As a result, for nearly three-quarters of all denied non-group qualified health plan claims, the reason is unclear. Denial rates vary by state: Mississippi and Indiana patients experienced the highest denial rates; insurers there denied 29% of all in-network claims. Only 1% of ACA Marketplace plan enrollees appealed their denied claims, and of those, nearly two-thirds (63%) were still denied coverage at the end of the appeal process. Pollitz et al., “Claims Denials and Appeals in ACA Marketplace Plans in 2020,” Kaiser Family Foundation, 7/05/2022
U.S. administrative costs once again prove high: Billing and insurance-related costs for inpatient bills ranged from $6 in Canada to $215 in the U.S., according to a microlevel accounting study of 5 nations. Australia, which has a mix of public and private payers, was similar to the U.S. The other nations included in the study were Germany, the Netherlands, and Singapore. Richman et al., “Billing and Insurance-Related Administrative Costs: A Cross-National Analysis,” Health Affairs, August 2022
Blues not paying taxes: A dozen Blue Cross Blue Shield (BCBS) insurers have not paid any net federal taxes since 2017 when Congress repealed the alternative minimum tax, while the government has refunded $6.6 billion to those insurers. Meanwhile, a federal judge approved a $2.67 billion antitrust settlement against 34 BCBS plans. Herman, “Many Blue Cross Blue Shield plans aren’t paying taxes — and instead are swimming in refunds,” STAT News, 6/15/2022; Tepper, “Judge approves Blue Cross Blue Shield $2.67B antitrust settlement,” Modern Healthcare, 8/9/2022
Cigna profits up: Cigna’s net income rose 6.2% to $1.5 billion for the second quarter of 2022, driving the company’s medical loss ratio (MLR) to 80.7%, compared with 84.4% for the same period in 2021. Reduced emergency department and surgery utilization drove the decline in spending, while Cigna also repriced its government-sponsored business to lower its MLR. Tepper, “Low medical spending drove Cigna’s quarterly profit,” Modern Healthcare, 8/4/22
Diabetes patients in Medicare Advantage plans have worse health: Type 2 diabetes patients on Medicare Advantage (MA) plans are more likely to have worse health than those in Traditional Medicare (TM), with MA patients having statistically significant higher systolic blood pressure (+0.2 mmHg) and worse blood glucose control (+0.1% A1C). While MA patients were more likely to receive preventive treatment, they were less than likely than TM patients to receive newer, more expensive treatments like SGLT2 inhibitors (5.4% in MA vs. 6.7% in TM) and GLP-1 receptor agonists (6.9% in MA vs. 9.0% in TM). Essein et al., “Diabetes Care Among Older Adults Enrolled in Medicare Advantage Versus Traditional Medicare Fee-For-Service Plans: The Diabetes Collaborative Registry,” Diabetes Care, 7/6/2022
Blues expand their Medicare Advantage market: Health Care Service Corp. (HCSC) plans its largest expansion ever into Medicare Advantage (MA). HCSC – which sells Blue Cross and Blue Shield plans in Illinois, Montana, New Mexico, Oklahoma, and Texas – plans to expand its MA business into 150 new counties for 2023. The previous year, HCSC’s MA plans expanded to 90 new counties, increasing membership by 10,000. Tepper, “HCSC doubles down on Medicare Advantage as market share declines,” Modern Healthcare, 8/3/2022
Phantom docs pervade Medicaid managed care: Only one-third of mental health prescribers listed in Oregon’s Medicaid directories provided care to Medicaid patients in 2018. Nearly six in ten providers (58.2%) in network directory listings were “phantom” providers who did not see Medicaid patients, including 67.4% of mental health prescribers. Zhu et al., “Phantom Networks: Discrepancies Between Reported and Realized Mental Health Care Access in Oregon Medicaid,” Health Affairs, July 2022
NHS outsourcing associated with more deaths: Outsourcing in the U.K.’s National Health Service to private, for-profit providers corresponds with an increase in treatable mortality and a decline in the quality of care, resulting in an additional 557 deaths between 2014 and 2020. From 2013 to 2020, outsourcing grew from 3.9% to 6.4%, with £11.5 billion given to private companies. Each 1% annual increase in outsourcing spending was associated with a 0.38% increase in treatable mortality (0.29 deaths per 100,000). Goodair & Reeves, “Outsourcing health-care services to the private sector and treatable mortality rates in England, 2013-20: An observational study of NHS privatisation,” The Lancet Public Health, July 2022
Health industry profits set to increase: McKinsey & Company estimates health care earnings will rise by 6% each year between 2021 and 2025, resulting in $31 billion in profits for the health industry. Increased profits are expected to come from government programs like Medicare Advantage and Medicaid managed care, and increased demand for non-acute care. Meanwhile, HCA Healthcare told investors they expect to pass along rising costs to commercial insurers through price negotiations, while UnitedHealth Group indicated receptiveness to higher prices. Singhal & Patel, “The future of US healthcare: What’s next for the industry post-COVID-19,” McKinsey & Company, 7/19/2022; Muoio, “Health Systems Confident Payers Will Concede Higher 2023 Rates,” Fierce Healthcare, 7/29/2022
CEOs cash in: The chief executives of approximately 300 health care companies reaped $4.5 billion in 2021, with an average CEO receiving $15.3 million, according to a STAT analysis. The highest paid CEOs usually come from pharmaceutical and medical device companies. Regeneron CEO Leonard Schleifer took in $453 million, or 10% of the total. Salaries make up less than 6% of pay, while realized gains of stock awards compose the greatest portion of pay packages. By comparison, average U.S. household income is $67,000. The $4.5 billion total could provide health insurance to 580,000 individuals for one year. Herman et al., “Health care’s high rollers: As the pandemic raged, CEOs’ earnings surged,” STAT News, 7/18/2022
ED facility fees higher at for-profit hospitals: High-acuity self-pay patients who visited for-profit emergency departments (EDs) were charged an average of $1,218 more than similar patients at non-profit EDs in 2021. Higher facility fees were also charged at hospitals with more than 250 beds (by $826) and system-affiliated EDs (by $311). EDs in high-poverty areas charged $450 less, on average, than EDs in affluent communities. Henderson & Mouslim, “Hospital and Regional Characteristics Associated with Emergency Department Facility Fee Cash Pricing,” Health Affairs, July 2022
Amazon continues foray into health care: Tech giant Amazon will acquire boutique primary care chain One Medical for $3.9 billion. One Medical has 767,000 enrolled patients. Amazon’s health care business includes a virtual and in-home urgent care service and an online pharmacy. Amazon previously acquired PillPack for $753 million in 2018 to jump start its pharmacy business. Lerman & Shaban, “Amazon will see you now: Tech giant buys health-care chain for $3.9 billion,” Washington Post, 7/21/2022
Private Equity draws attention for foray into hospice and autism care: Private Equity (PE) ownership of hospice agencies increased from 106 (3.4% of total hospices) in 2011 to 409 (7.3%) in 2019. Nonprofits represented 72% of hospices acquired by PE in that time. PE is also entering the child autism therapy business, alarming parents, clinicians, and experts. They say PE investments in Applied Behavior Analysis therapy has degraded the quality of service, turning it into “fast food therapy” that could even be harmful for children. Hawryluk, “Hospices Have Become Big Business for Private Equity Firms, Raising Concerns About End-of-Life Care,” Kaiser Health News, 7/29/2022; Bannow, “Parents and clinicians say private equity’s profit fixation is short-changing kids with autism,” STAT News, 8/15/2022
Unions growing among younger physicians: Coinciding with an increase in union organizing nationally, the Committee of Interns and Residents reports its membership has grown by 37.5% since 2019, adding 6,000 new members to bring its current membership to 22,000. Over the past two years, the Union of American Physicians and Dentists has grown by 9.9% and now represents 5,000 professionals. Organizers say the growth is driven by concern over personal protective equipment and worker and patient safety during the Covid-19 pandemic, along with the increasing number of physicians who are directly employed by health systems rather than practicing independently. Christ, “More physicians seek unions amid changing landscape, COVID-19,” Modern Healthcare, 7/20/2022
New drugs, new high prices: The average cost of newly launched drugs increased by 20% per year from 2008 ($2,115) to 2021 ($180,007). Even after adjusting for manufacturer discounts, prices rose by 11% each year. At the same time, a greater proportion of new drugs are high priced, with 47% costing $150,000 or more in 2020 and 2021, compared with 9% of drugs launched from 2008 to 2013. So far this year, the median annual price of 13 new novel drugs for chronic conditions is $257,000. Rome et al., “Trends in Prescription Drug Launch Prices, 2008-2021,” JAMA, 6/7/2022; Beasley, “Newly launched U.S. drugs head toward record-high prices in 2022,” Reuters, 8/16/2022
Bias pervades drug effectiveness studies: Drug, medical device, and biotech industry-sponsored cost effectiveness analyses (CEAs) are twice as likely to report a treatment as cost effective compared with independently conducted CEAs. Incremental cost effectiveness ratios (ICERs) from industry-sponsored CEAs were 33% lower than from non-industry studies. Treatments with lower ICERs are more likely to receive insurance coverage approval. Xie, “Industry sponsorship bias in cost effectiveness analysis: registry based analysis,” BMJ, 6/22/2022
More industry bad behavior: Biogen agreed to pay $900 million to settle a whistleblower case alleging the company paid kickbacks to physicians for its multiple sclerosis drug and disguised marketing programs as educational sessions. Meanwhile, AbbVie was reprimanded by a U.K. pharmaceutical trade group for code of conduct violations. The Prescription Medicines Code of Practice Authority criticized the company’s sales reps for “strategic loitering” and circumventing hospital Covid-19 non-essential visitor restrictions. Silverman, “Biogen agrees to pay $900 million ot settle whistleblower case alleging kickbacks and sham speaking events,” STAT News, 7/20/2022; Silverman, “AbbVie is scolded by a trade group over sales rep ‘strategically loitering’ in a hospital,” STAT News, 7/27/2022
Medicare increases payments to hospitals following key lobbying: Hospitals will receive a 4.3% increase in payments for inpatient services in 2023, the largest rate increase in 25 years. This follows lobbying from hospital groups after the Centers for Medicare and Medicaid Services proposed a 3.2% increase. Medicare will also increase payment rates for hospices (3.8%), inpatient rehabilitation (3.2%), and inpatient psychiatric services (2.5%). Herman, “Hospitals win higher payments from Medicare after lobbying campaign,” STAT News, 8/1/2022; Goldman, “CMS hikes Medicare pay for rehab, psychiatric and hospice providers,” Modern Healthcare, 7/27/2022
“The maternal mortality consequences of losing abortion access,” by Amanda Jean Stevenson, Leslie Root, Jane Menken, SocArVix Papers, 6/29/2022. “After the first year of no abortion occurring, we estimate increased exposure to the risks of pregnancy would cause an increase of 210 maternal deaths per year (24% increase), from 861 to 1071. The increase would be greatest among non-Hispanic Black people, for whom it would be 39%.”
“Universal healthcare coverage and health service delivery before and during the COVID-19 pandemic: A difference-in-difference study of childhood immunization coverage from 195 countries,” by Sooyoung Kim,Tyler Y. Headley,Yesim Tozan, PLOS Medicine, 8/16/2022. “We observed that countries with greater progress toward UHC [Universal Health Coverage] were associated with significantly smaller declines in childhood immunization coverage during the pandemic. This identified association may potentially provide support for the importance of UHC in building health system resilience. Our findings strongly suggest that policymakers should continue to advocate for achieving UHC in coming years.”
“Trends in Out-of-Pocket Costs for Naloxone by Drug Brand and Payer in the US, 2010-2018,” by Evan D. Peet, Ph.D.; David Powell, Ph.D.; Rosalie Liccardo Pacula, Ph.D., JAMA Health Forum, 8/19/2022. “This observational study of 719,612 pharmacy claims data shows that OOP costs of naloxone grew substantially beginning in 2016. However, OOP costs did not increase for all patients and all brands of naloxone but primarily for uninsured patients and for the Evzio brand.”
“Association of Chronic Disease With Patient Financial Outcomes Among Commercially Insured Adults,” by Nora V. Becker, M.D., Ph.D.; John W. Scott, M.D., M.P.H.; Michelle H. Moniz, M.D., M.Sc.; Erin F. Carlton, M.D., M.Sc.; John Z. Ayanian, M.D., M.P.P., JAMA Internal Medicine, 8/22/2022. “This cross-sectional study of commercially insured adults linked to patient credit report outcomes shows an association between increasing burden of chronic disease and adverse financial outcomes.”
“CMS Should terminate the Medicare Advantage Program,” by Physicians for a National Health Program, 8/25/2022. “It would be far more cost-effective for CMS to improve traditional Medicare by capping out-of-pocket costs and adding improved benefits within the Medicare fee-for-service system than to try to indirectly offer these improvements through private plans that require much higher overhead and introduce profiteers and perverse incentives into Medicare, enabling corporate fraud and abuse, raising cost to the Medicare Trust Fund, and worsening disparities in care. These problems are not correctable within the competitive insurance business model, and the Medicare Advantage program should be terminated.”
“Uncovered Medical Bills after Sexual Assault,” by Samuel L. Dickman, M.D.; Gracie Himmelstein, M.D., Ph.D.; David U. Himmelstein, M.D.; Katherine Strandberg, M.P.A.; Alecia McGregor, Ph.D.; Danny McCormick, M.D.; Steffie Woolhandler, M.D., M.P.H., The New England Journal of Medicine, 9/15/2022. “Our findings indicate that an estimated 17,842 persons who sought emergency department care related to sexual assault [out of 112,844 such visits in 2019] were expected to pay the often-substantial costs themselves. Other data indicate that even privately insured sexual assault victims pay, on average, 14% of emergency department costs out-of-pocket.”
“Prevalence and Risk Factors for Medical Debt and Subsequent Changes in Social Determinants of Health in the US,” by David U. Himmelstein, M.D.; Samuel L. Dickman, M.D.; Danny McCormick, M.D., M.P.H.; David H. Bor, M.D.; Adam Gaffney, M.D., M.P.H.; Steffie Woolhandler, M.D., M.P.H., JAMA Network Open, 9/16/2022. “In this cross-sectional and cohort study of survey data from 2017 to 2019, 10.8% of adults carried medical debt, including 10.5% of the privately insured, and 9.6% of residents of Medicaid-expansion states, significantly fewer than in non-expansion states. Over 3 years, decreases in health status and coverage loss were significant risk factors associated with acquiring medical debt, which was, in turn, associated with a significant 1.7-fold to 3.1-fold higher risk of worsening housing and food security.”

In California, Dr. Ana Malinow organized an action outside the Federal Building in San Francisco to both celebrate the 57th anniversary of Medicare being signed into law and to warn against creeping privatization of the program through schemes like Direct Contracting and REACH. The event, titled “Make it a Birthday, Not a Funeral,” was emceed by Dr. Corinne Frugoni and sponsored by Senior Disability Action, the California Alliance for Retired Americans, and DSA San Francisco, among other organizations. In terms of online activism, the California chapter launched its new website (pnhpca.org), developed in large part by chapter co-chair Dr. Kathleen Healey, and celebrated the release of a health care savings calculator from Healthy California Now (healthyca.org/calculator), which benefited from the contributions of Drs. Hank Abrons and Jim Kahn.
To get involved in California, contact Dr. David Leibowitz at dleibow@gmail.com.
In Georgia, chapter leaders renewed their efforts to engage with members of the Atlanta City Council in support of a municipal Medicare-for-All resolution. Leaders also reached out to Sens. Raphael Warnock and John Ossoff to thank them for supporting a pair of ultimately unsuccessful amendments to the Inflation Reduction Act that would have extended dental, vision, and hearing coverage to Medicare patients and basic health coverage to residents of states that have not expanded Medicaid.
To get involved in Georgia, contact Dr. Liz McCord at pnhpgeorgia@gmail.com.

In Illinois, members of PNHP and the Northwestern University Students for a National Health Program (SNaHP) chapter joined ONE Northside in a spirited demonstration outside Centene’s Chicago office. They demanded that the insurance company stop its fraudulent denial of claims for being “out of network,” highlighting the case of a community member who was wrongfully billed $999 for routine blood work and who had spent countless hours challenging the error. Ultimately, activists demanded that commercial insurance companies be replaced by a health care system that is publicly financed, nonprofit, and fully accountable to the public: improved Medicare for All.
To get involved in Illinois, contact Dr. Monica Maalouf at mmaalouf88@gmail.com.
In Kentucky, chapter members participated in a Continuing Medical Education (CME) program sponsored by the Kentucky Medical Association titled, “The U.S. Healthcare Delivery System: Where it Succeeds, How it Fails to Meet the Needs of Patients and Providers, and Options for Change.” The program was organized by Dr. Susan Bornstein and took place over Zoom on August 17. On July 30, members celebrated Medicare’s 57th birthday by distributing flyers urging an end to Direct Contracting and REACH at Louisville farmers’ markets, and at a Madison County picnic.
To get involved in Kentucky, contact Kay Tillow at nursenpo@aol.com or Dr. Garrett Adams at kyhealthcare@aol.com.
PNHP’s Maine chapter, Maine AllCare, has formed a new 501(c)(4) organization, HealthCare for All Maine, that will engage in lobbying efforts to bolster single-payer legislation. A team of activists within the chapter has also formed a Physician Working Group that is focused on messaging to medical professionals and updating the Maine Medical Association’s position on single payer.
To get involved in Maine, contact Karen Foster at kfoster222@gmail.com.
In Minnesota, a group of 17 rising 2nd-year medical students and graduate students working towards their MPH participated in the chapter’s Summer Education Program. Seven of these students completed individual projects as part of a paid internship, and shared them at the PNHP Minnesota Annual Summer Picnic on August 12. Activists also joined forces with Health Care for All Minnesota to table at the Twin Cities Pride Festival in June and at the Minnesota Farmfest in early August. In late August and early September, more than 50 volunteers tabled at the Minnesota State Fair, spreading the word about single payer to crowds totalling over two million for the week.
To get involved in Minnesota, contact Jen Crawford at pnhpminnesota@gmail.com.

In New Hampshire, chapter leaders worked with state legislators to explore a bill that would form a multi-state single-payer compact, seeking power in numbers and collaboration among activists and legislatures seeking to pursue state-level initiatives. Physician members also made presentations to the SNaHP chapter at the Geisel School of Medicine at Dartmouth.
To get involved in New Hampshire, contact Dr. Donald Kollisch at donald.o.kollisch@dartmouth.edu.
The New Jersey Universal Healthcare Coalition finalized plans to collaborate with Rutgers University on a poll of voters across the state, seeking to gauge their opinion of our current health care “system” and assess their enthusiasm for single-payer reform. Several members are also planning to present resolutions to the Medical Society of New Jersey.
To get involved in New Jersey, contact Dr. Lloyd Alterman at lloydalterman52@gmail.com.
In New York, PNHP’s New York-Metro chapter announced the hiring of a new Executive Director, Morgan Moore, who has been instrumental to the growth of the chapter in recent years. When the Covid-19 pandemic hit, she played a major role in transitioning chapter activities online and continuing the series of high-quality monthly educational forums at PNHP NY Metro. She also launched the chapter’s #MedStoryMonday social media campaign, where health workers are encouraged to share their personal stories of how the for-profit health insurance system has negatively impacted their ability to provide care. Morgan started as Executive Director September 1 and Mandy Strenz, who had been serving as Acting Executive Director, returned to her role as Chapter Coordinator. Earlier in the summer, the NY Metro chapter collaborated with other local advocacy groups to celebrate the anniversary of Medicare and Medicaid. Members presented oversized birthday cards to the offices of Sens. Gillibrand and Schumer, urging them to fight back against profiteering by ending Medicare Direct Contracting and REACH.
To get involved in New York, contact Morgan Moore at morgan@pnhpnymetro.org.


Health Care Justice – North Carolina in Charlotte celebrated Medicare’s birthday by delivering sheet cakes decorated with faux Medicare cards, balloons, and information about Direct Contracting and REACH to local Congressional offices. The chapter also developed a two-page letter containing information about PNHP and Medicare for All (available at healthcarejusticenc.org) which they encouraged members to print and deliver to their health care providers during office visits. On August 21, members continued their annual tradition of marching in the Charlotte Pride Parade.
To get involved in Health Care Justice-NC, contact Dr. George Bohmfalk at gbohmfalk@gmail.com or Dr. Jessica Schorr Saxe at jessica.schorr.saxe@gmail.com.
Members of Healthcare For All – Western North Carolina in Asheville held a public downtown rally to celebrate the 57th anniversary of Medicare. Activists brought banners, gift bags, and sidewalk chalk for visitors to write big, bold messages about what Medicare means to them and why it needs to be protected from profiteers. Chapter leaders also held a well-attended informational meeting at a local retirement community where they screened “FIX IT” and fielded many concerned questions about Direct Contracting and REACH; similar events will be held on a monthly basis going forward.
To get involved in Health Care for All WNC in Asheville, contact Terry Hash at theresamhash@gmail.com.
In Vermont, students who participated in the Northern New England online internship program continued their single-payer activism by giving presentations to a variety of groups, including the League of Women Voters’ National Convention and One Payer States.
To get involved in Vermont, contact Dr. Betty Keller at bjkellermd@gmail.com or Ted Cody tscody@vermontel.net.
The PNHP Washington chapter worked closely with Puget Sound Advocates for Retirement Action, Health Care is a Human Right WA, and other progressive organizations to protest the ongoing privatization of Medicare. On July 29, the day before Medicare’s 57th birthday, members of this coalition joined forces to rally and picket outside the regional office of the Dept. of Health and Human Services in Seattle, demanding an end to Direct Contracting and REACH and eventually securing a meeting with the Regional Director of HHS. In early August, PNHP-WA co-sponsored a “Righteous Mothers” benefit concert to stop the privatization of Medicare, during which Medicare-for-All Act lead sponsor Rep. Pramila Jayapal addressed the crowd.
To get involved in Washington, contact pnhp.washington@gmail.com.

In West Virginia, chapter members collaborated with five local health activist groups to plan and host a “Happy Birthday Medicare and Medicaid” event in Charleston on July 30. The event took place at a Federally Qualified Health Center, and the chapter continued sending letters to similar FQHC providers throughout the state telling them about PNHP and inviting them to join. Chapter leaders also drafted an anti-REACH resolution that was eventually passed by the West Virginia Democratic Party, and sent letters to Gov. Justice and all state legislators opposing any legislation that criminalizes health care providers and patients for providing or receiving abortion services.
To get involved in West Virginia, contact Dr. Dan Doyle at pnhp.wv@gmail.com.

“Prescription for Healthcare: Threats to Medicare,” WFHB Community Radio, 9/05/2022, featuring Dr. Ana Malinow
Click the links below to jump to different sections of the newsletter. To view a PDF version of the shorter print edition of the newsletter, click HERE.
If you wish to support PNHP’s outreach and education efforts with a financial contribution, click HERE.
If you have feedback about the newsletter, email info@pnhp.org.
After several months of intense educating and organizing by PNHP and our allies, the Centers for Medicare and Medicaid Services (CMS) announced the termination of the controversial Medicare Direct Contracting model in February, admitting the program “did not align” with the Biden administration’s vision. At the same time, CMS said it planned to replace DC with a nearly identical program called “ACO REACH.”
CMS’ “rebranding” of Direct Contracting to REACH taught PNHP some important lessons. First, we knew that our work made an enormous impact — CMS would have never canceled the program if not for our campaign. But it wasn’t enough. We learned that we could never end Medicare profiteering unless we organize a powerful, national, grassroots movement.
To meet that challenge, PNHP expanded our organizing, working with chapter leaders to give dozens of presentations to community and senior groups. Along with our allies, we helped pass anti-DC/REACH resolutions in the Seattle City Council and the Arizona Medical Association.
The organizing paid off. On May 23, PNHP hosted our biggest event ever: The launch of our “Summer of Action” against Medicare profiteering. More than 3,000 activists participated in this online event, where they learned the nuts-and-bolts of Direct Contacting and REACH, heard powerful testimonials from Medicare beneficiaries, and were inspired to take action by Congresswomen Katie Porter and Pramila Jayapal.
Anyone can get involved in our Summer of Action against Medicare profiteering. Here’s how:
Go to ProtectMedicare.net to find campaign updates, sample scripts, fact sheets, videos and more.
Medicare advocates are celebrating two big victories in the movement to protect Medicare from profiteering and privatization.
In Seattle, seniors from the Puget Sound Advocates for Retirement Action, along with PNHP’s Washington Chapter, proposed a resolution against Medicare Direct Contracting and REACH. The resolution, which demands that the Dept. of Health and Human Services and President Biden immediately end Direct Contracting/REACH and protect Medicare from profiteering, was introduced by Councilmember Teresa Mosqueda and passed unanimously on April 26.
That same week, PNHP members Dr. Eve Shapiro and Dr. Michael Hamant introduced and successfully passed a similar resolution at the annual meeting of the Arizona Medical Association.

Drs. Shapiro and Hamant explained to their colleagues that since most physicians are now employed by large groups or health systems, they may find themselves practicing in Direct Contracting Entities (DCEs) without their knowledge or consent. Among concerns cited by the resolution is that “DCEs are allowed to keep as profit and overhead what they don’t pay for in health services, therefore giving them a dangerous financial incentive to restrict seniors’ care.”
The Seattle and Arizona resolutions are part of PNHP’s strategy to expand the fight against Medicare profiteering into every state and Congressional district in the nation.

After years of education and organizing by PNHP members, the 25,000-member American Public Health Association (APHA) strongly endorsed a Medicare for All policy at its most recent annual meeting, concluding that, “The most equitable and cost-effective health care system is a public, single-payer system.”
The policy statement was crafted by a working group of the APHA’s Medical Section, which included PNHP leaders Drs. Anthony Spadaro, Oli Fein, and Gordy Schiff, working with colleagues in the epidemiology, public health, pharmacy, and social work professions.
The working group published the position paper, “The American Public Health Association Endorses Single-Payer Health System Reform,” in the June 2022 edition of Medical Care, the official journal of the Medical Care Section of the APHA.
“The APHA is our nation’s strongest voice for public health, and I’m thrilled that my colleagues came together to support single payer as the solution to our fragmented and profit-oriented health system,” said Dr. Spadaro, a resident emergency medicine physician at the University of Pennsylvania and former board member of Students for a National Health Program.
The APHA resolution is part of PNHP’s Medical Society Resolutions campaign, which seeks to organize the medical profession by passing Medicare-for-All resolutions in every medical society in the U.S. The MSR campaign has passed resolutions in the American College of Physicians, the Society of General Internal Medicine, and the Hawaii, Vermont, New Hampshire and Washington state medical societies.
For more information on the PNHP Medical Resolutions Campaign, visit MedicalSocietyResolutions.org.

PNHP is known for its independence and unwavering commitment to a gold standard in health policy, in large part because of how we’re funded — by dues and donations of our members, never with corporate money that could compromise our mission.
This spring, longtime PNHP member Dr. Rob Stone discovered a new way to support PNHP’s mission: Through a generous gift of stock.
Dr. Stone, a palliative medicine physician, lives with his wife Karen in Bloomington, Indiana. He’s been a PNHP member since 2000, including roles as a board member and advisor, and usually donates about $1,000 per year to PNHP.
But when he turned 70 earlier this year, Dr. Stone began thinking more about his retirement and his legacy in the health justice movement. He’d been especially excited about PNHP’s campaign to stop Medicare privatization through Direct Contracting, and how important that was for the future of Medicare for All.
As he thought about how he could better support PNHP, Dr. Stone inherited Microsoft stock from his mother, who bought it in the 1980s; the stock had appreciated in value to about $40,000.
“I did the math and realized that if I sold the stock and kept it, I’d have to pay about $5,000 in capital gains taxes,” said Dr. Stone. “But if I donated the stock to PNHP, I would get a $40,000 tax deduction.”
What was more important was the incredible impact that his gift could make to PNHP’s mission.
“I’ve been very excited about the work that PNHP has been doing over the past year and was thrilled that I had an opportunity to help keep that momentum going,” Dr. Stone added. “My mother always encouraged me to give and to lead by example, and I’m hoping that my gift of stock will encourage other PNHP members to do the same.”
Part of Dr. Stone’s stock gift will support the Dorothy W. Stone Scholarship to help medical students attend PNHP’s Annual Meeting in November.
“I strongly encourage other members to think about donating stock to PNHP,” said Dr. Stone. “It’s easy. You get huge tax benefits, and you can make Medicare for All part of your legacy.”
Nominations for PNHP’s Board of Directors will be open from August 1 to 31, with seats up for election in all regions and for at-large representation. Recent bylaws changes expands the number of seats on the Board, so that more members have the opportunity to serve on PNHP’s leadership team. The Board invites nominations and applicants from members interested in contributing to a diverse Board of Directors.
The following seats up for election for 2-year terms:
Questions about qualifications and expectations should be sent to deputy director Matthew Petty at matt@pnhp.org. Nominations (by self or others) are due to matt@pnhp.org by August 31, 2022. Ballots for electronic voting will be circulated in September 2022; please make sure your current email address is on the file with PNHP’s national office.
Americans forego care due to cost: More than half of adults (51%) report that in the past year, they have delayed or gone without medical services due to costs, including 35% who put off dental services, 25% who put off vision care, and 24% who delayed general visits to their health provider. Kirzinger et al., “Health Tracking Poll March 2022: Economic concerns and health policy, the ACA, and views of long-term care facilities,” Kaiser Family Foundation, 3/31/2022
Cancer patients go into debt for care: More than half (51%) of U.S. cancer patients have gone into debt to cover the cost of care. Of those who incurred debt, 53% faced collections and 46% saw their credit scores drop; 62% have since delayed or avoided medical care, while half have sought the least expensive treatment options due to debt. In order to pay for cancer care, more than a quarter (28%) of patients depleted most or all of their savings, 28% have gone into credit card debt, 20% borrowed money from family and friends, and 11% took out another type of loan, like a payday loan or home refinancing. More than a third of cancer patients (36%) cut back on food, clothing, and basic household expenses. “Survivor Views: Cancer & Medical Debt, February 2022 Survey Findings Summary,” American Cancer Society, 3/17/2022
Women in the U.S. face worse health care and outcomes than peer countries: Nearly half (49%) of women of reproductive age in the U.S. skip or delay care because of costs, a rate more than double that of most peer nations. More than half (52%) of women in the U.S. report problems paying medical bills, compared to 10% in the U.K. Over one-quarter (27%) of American women spent $2,000 or more in out-of-pocket medical costs, as compared with less than 5% percent of women in the U.K., France, and Netherlands, and less than 10% in Germany, New Zealand, Canada and Norway. American women have the highest rate of avoidable deaths, and the U.S. maternal mortality rate is three times higher than France and seven times higher than Germany. Gunja et al., “Health and Health Care for Women of Reproductive Age: How the United States Compares with Other High-Income Countries,” Commonwealth Fund, 4/05/2022
Americans lack access to primary care: Compared to a set of 10 other wealthy nations, Americans are the least likely to have a longstanding relationship with a primary care provider, least likely to have access to home visits by a primary care provider, and are the least likely to be able to see a provider after regular office hours. The U.S also has the largest income gap between generalist and specialist physicians ($236,000 vs. $526,000 per year) and the highest medical school tuition. FitzGerald, “Primary Care in High-Income Countries: How the United States Compares,” Commonwealth Fund, 3/15/2022
High costs keep Medicare beneficiaries from critical treatments: Medicare Part D beneficiaries who did not receive subsidies to cap or lower their out-of-pocket costs were nearly twice as likely to not fill prescriptions for serious health conditions, since Part D drug costs can reach $10,000 or more. Among patients without subsidies, 30% did not fill their prescriptions for cancer drugs, 22% did not fill prescriptions for hepatitis C, and more than 50% did not fill therapies for high cholesterol or immune disorders. Dusetzina et al., “Many Medicare Beneficiaries Do Not Fill High-Price Specialty Drug Prescriptions,” Health Affairs, April 2022
Disparities plague drug affordability in Medicare: Among Medicare beneficiaries, 3.5 million seniors (6.6%) and 1.8 million under-65 adults with disabilities (22.7%) had difficulty affording their medications in 2019. Hispanic/Latinx and Black seniors were roughly 1.5 times more likely to have affordability problems compared to white seniors, and two times as likely not to get needed prescriptions due to cost. Among beneficiaries with diabetes, 10% of seniors and 26% of under-65 disabled adults reported medication affordability problems. Tarazi et al., “Prescription Drug Affordability among Medicare Beneficiaries,” U.S. Department of Health and Human Services Assistant Secretary for Planning and Evaluation, Office of Health Policy, 1/19/2022
Major racial disparities among cancer patients with COVID-19: Black cancer patients who were infected with COVID-19 had worse outcomes than similar white patients, with higher rates of hospitalization, intensive care unit admission, and mechanical ventilation. Black patients also experienced higher rates of lung, heart, and vascular complications, acute kidney injuries, and all-cause mortality, and were less likely to be treated with remdesivir and more likely to be treated with hydroxychloroquine. Fu et al., “Racial Disparities in COVID-19 Outcomes Among Black and White Patients with Cancer,” JAMA Network Open, 3/28/2022
Immigrants face major barriers to care: In 2020, more than one in four (42%) undocumented immigrants and 26% of documented immigrants were uninsured, compared to 8% of U.S. citizens. More than a quarter (28%) of undocumented children and 17% of documented children were uninsured, compared to 4% of children with citizen parents. The vast majority of immigrants (83%) were employed or lived with someone who was employed full-time (the same rate as citizens), but undocumented immigrants are not eligible for any kind of financial assistance through Medicaid coverage or tax credits. Income is also a barrier to care, as 44% of undocumented immigrants and 39% of documented immigrants are low-income, compared to 25% of citizens. “Health Coverage of Immigrants,” Kaiser Family Foundation, 04/06/2022
Diabetic amputations higher in states that didn’t expand Medicaid: Among patients of color that were hospitalized for diabetic foot ulcers in the two years after the implementation of the ACA, researchers found a 9% increase in major amputations in states that did not expand Medicaid, but no change in states that did expand. For uninsured adults, the amputation rate decreased 33% in expansion states but did not change in non-expansion states. Tan et al., “Rates of Diabetes-Related Major Amputations Among Racial and Ethnic Minority Adults Following Medicaid Expansion Under the Patient Protection and Affordable Care Act,” JAMA Network Open, 3/24/2022
Commercial insurers delay and deny care: Despite an agreement between the insurance industry and the AMA to streamline the prior authorization (PA) process, 84% of physicians report that the number of PAs required for prescriptions and medical services has increased over the last five years, with 65% saying that it is difficult to determine whether a prescription or medical service requires PA. Physicians report phone calls as the most common method for completing PAs (59%), with 45% of providers always or often using fax machines. An overwhelming majority (88%) of physicians report that PA interferes with continuity of care. “Measuring progress in improving prior authorization: 2021 Update,” American Medical Association, May 2022
High-deductible health plans (HDHPs) a major barrier to mental health care: When employers switched their employees into HDHPs, enrollees with depression were 18% less likely to seek outpatient care, those with ADHD were 15% less likely, and those with anxiety were 14% less likely. Inpatient hospital admissions also dropped significantly for HDHP enrollees with depression (19%), anxiety (16%), and ADHD (6%). After employers switched to HDHPs, overall plan spending for depression, anxiety, and ADHD dropped by $1,137, $984, and $868, respectively, but individual employees’ own spending increased by $326, $321, and $281, respectively. The switch to HDHPs also caused enrollees with anxiety or depression to skip preventative care such as breast, cervical, and prostate cancer screenings, as well as flu and pneumonia vaccinations. Fronstin and Roebuck, “How Do High-Deductible Health Plans Affect Use of Health Care Services and Spending Among Enrollees with Mental Health Disorders?” Employee Benefit Research Institute, 3/10/2022
Commercial insurers won’t pay for catheter supplies: Nearly 80% of catheter users with commercial insurance had to pay out-of-pocket (OOP) for catheters and catheter supplies, including 88% of those on UnitedHealthcare, 79% on BCBS, and 75% on Aetna, compared to 53% of those on public plans. Commercial plan enrollees paid more than three times the amount in OOP catheter expenses ($1,621) than those in public plans ($531). United Healthcare members faced the highest average OOP costs at $2,188 per year. “National Survey Among Catheter Users: A Study to Examine Catheter Usage and Catheter Coverage by Health Plan,” Spina Bifida Association and Duke Health, 3/16/2022
COVID survivors slammed with medical bills: Getting hospitalized for a serious case of COVID-19 left many commercially insured patients with bills averaging $1,600 to $4,000. More than one in ten patients (11%) with commercial insurance and 9.3% of patients with Medicare Advantage had more than $2,000 in bills in the first six months after a COVID-19 hospitalization. For patients hospitalized for pneumonia, OOP spending exceeded $2,000 for 12.1% with commercial insurance and 17.2% with Medicare Advantage plans. Chua, et al., “Out-of-Pocket Spending for Health Care After COVID-19 Hospitalization,” American Journal of Managed Care, 3/16/2022
Insurers celebrate record profits in early 2022: The six largest commercial insurers pocketed a combined $11.2 billion in the first three months of this year. UnitedHealth Group was the most profitable, reporting $5 billion in profit in the first quarter of 2022, followed by CVS Health (Aetna) with $2.3 billion, Anthem at $1.8 billion, Cigna at $1.2 billion, and Humana at $930 million. Minemyer, “UnitedHealth was this quarter’s most profitable payer—again,” Fierce Healthcare News, 5/9/2022
Insurance CEOs pocket millions in compensation: CEOs at the six largest commercial insurers earned nearly $115 million in combined total compensation last year. The late Michael Neidorff, former CEO of Centene, topped the list with $20.6 million in total compensation for 2021; followed by Karen Lynch of CVS Health (Aetna) at $20.4 million; Gail Boudreaux of Anthem with $19.3 million; David Cordani of Cigna with $19.9 million; Andrew Witty of UnitedHealth Group with $18.4 million; and Bruce Broussard of Humana with $16.5 million. Minemyer, “Centene’s Michael Neidorff was the highest-paid payer CEO last year. Take a look at what other execs earned,” Fierce Healthcare News, 4/27/2022
Commercial insurers drive up federal health spending: In 2020, commercial insurers’ overhead totaled $301.4 billion, up from $236.6 billion in 2019. Commercial Medicare Advantage plans accounted for $63.4 billion of that total, up 41.2% from 2019. The overhead of commercial insurers who run Medicaid managed care plans was $55.5 billion, up 64.9% from 2019. Hartman et al., “National Health Care Spending In 2020: Growth Driven By Federal Spending In Response To The COVID-19 Pandemic,” Office of the Actuary, CMS, published in Health Affairs, 12/21/2021
Medicare Advantage (MA) spending drags down Medicare budget in pandemic: Due to the sharp drop in utilization, 2020 spending on Part A and B services for Traditional Medicare (TM) decreased 5.8% from 2019, the first time annual spending has declined in more than 20 years. However, total Medicare spending increased because the federal government increased payments to commercial MA plans by 6.9%, since MA payments were determined in mid-2019 and not adjusted to reflect lower utilization. Biniek et al., “Traditional Medicare Spending Fell Almost 6% in 2020 as Service Use Declined Early in the COVID-19 Pandemic,” Kaiser Family Foundation, 6/1/2022
Medicare watchdog slams inflated Medicare Advantage costs: In 2020, Medicare paid 4% more for beneficiaries enrolled in MA than it would have if those beneficiaries were in Traditional Medicare. Medicare overpaid MA insurers by $12 billion just from upcoding alone. According to MedPAC, “The MA program has been expected to reduce Medicare spending since its inception … but private plans in the aggregate have never produced savings for Medicare, due to policies governing payment rates to MA plans that the Commission has found to be deeply flawed.” The Commission also noted that, “These policy flaws diminish the integrity of the program and generate waste from beneficiary premiums and taxpayer funds.” “Report to the Congress: Medicare Payment Policy,” Medicare Payment Advisory Commission, 3/15/2022
Medicare Advantage plans use prior authorization (PA) to deny needed care: An HHS watchdog found that among commercial MA plans’ PA denials, 13% were for services that met Medicare coverage rules and that the denials likely prevented or delayed necessary care. They also found that 18% of the MA plans’ denied payment requests met Medicare coverage rules and MA billing rules, and were thus improperly denied. “Some Medicare Advantage Organization Denials of Prior Authorization Requests Raise Concerns About Beneficiary Access to Medically Necessary Care,” U.S. Dept. of Health and Human Services Office of the Inspector General, 4/28/2022
Nearly half of seniors don’t understand limitation of MA plans: In a new survey, more than one-third (35%) of Medicare Advantage (MA) enrollees mistakenly believe they don’t have to stay in-network for care, while another 11% weren’t sure, and only half (50%) of MA enrollees said they understood that they don’t have free choice of provider. Grunebaum, “8 in 10 Rate Understanding of Medicare Advantage Good or Very Good: Survey,” MedicareGuide.com, 5/08/2022
Rampant health disparities among Medicare Advantage enrollees: Black, Indigenous and Alaska Native patients experienced the most significant disparities in clinical care among Medicare Advantage enrollees in 2021. American Indian and Alaska Native enrollees ranked lowest among all demographic groups for breast cancer screenings, respiratory conditions, and diabetes care. Compared to all MA enrollees, Black enrollees were less likely to receive follow-up care after emergency department visits for mental and behavioral health events, and also faced the most adverse prescribing practices, with clinicians more likely to dispense medications with significant side effects to Black people. Martino et al., “Disparities in health care in Medicare Advantage by race, ethnicicty and sex,” CMS Office of Minority Health, April 2022
High rate of “ghost” physicians in privatized Medicaid: In a study of four states from 2015 to 2017, researchers found that Medicaid managed care (or MMC, run by commercial insurers) provider network directories overstate how many physicians actually offer care to Medicaid enrollees. They found that 16% of adult primary care physicians listed in MMC networks qualified as “ghost physicians,” meaning they did not file any Medicaid claims in a year, and almost a third of MMC outpatient primary care and specialist physicians saw less than 10 Medicaid patients a year. Psychiatry was the specialty most likely to include ghost physicians, with 35% of MMC-contracted psychiatrists not seeing any Medicaid patients. Among all MMC-contracted providers, 25% of primary care doctors delivered 86% of the care, while 25% of specialists on average provided 75% of the care. Ludomirsky et al., “In Medicaid Managed Care Networks, Care Is Highly Concentrated Among a Small Percentage of Physicians,” Health Affairs, May 2022
Surge in private equity (PE) in health care: Total PE investment in the health care industry has increased 20-fold, from $5 billion annually in 2000 to $100 billion in 2018. Annual PE acquisitions grew from 78 in 2000 to 855 in 2018. Appelbaum and Batt, “Private Equity Buyouts in Healthcare: Who Wins, Who Loses?” Institute for New Economic Thinking Working Paper Series, No. 118, May 2020
Private equity (PE) acquisition of hospitals leads to reduced staffing, higher profits: Between 2005 and 2014, hospitals acquired by PE firms saw a 1.78 percentage point increase in operating margins, along with a 2.79% decrease in bed count (about 4.43 beds). PE acquisition also reduced full-time equivalents (FTEs) staffing by 5.05%, an average loss of 36.97 FTE staff, with total nursing FTEs reduced by 4.38% or 10.52 FTE nurses. The ratio of outpatient to inpatient visits also decreased by 4.58%, indicating an increase in inpatient utilization likely due to more aggressive price negotiation for inpatient care with commercial insurers. Cerullo et al., “Financial Impacts And Operational Implications Of Private Equity Acquisition Of US Hospitals,” Health Affairs, April 2022
Higher nursing staff levels save lives: In a study of over 700,000 Medicare beneficiaries with sepsis, researchers found that an increase in registered nurse hours per patient day was associated with a 3% decrease in 60-day mortality, suggesting that hospitals that provide more RN hours of care could likely decrease sepsis deaths. Cimiotti et al., “Association of Registered Nurse Staffing With Mortality Risk of Medicare Beneficiaries Hospitalized With Sepsis,” JAMA Health Forum, 5/27/2022
Nursing home unions save lives: Nursing home labor unions were associated with 10.8% lower resident COVID-19 mortality rates, and 6.8% lower worker COVID-19 infection rates. Researchers estimate that 8,000 fewer resident deaths would have occurred if all nursing home staff were unionized during the pandemic, since labor unions were associated with better infection control policies and COVID-19 outcomes for essential workers. Dean et al., “Resident Mortality and Worker Infection Rates from COVID-19 Lower in Union Than Nonunion U.S. Nursing Homes, 2020–21,” Health Affairs, 4/20/2022
Investor-owned hospitals push more low-value care: “Low-value care” refers to medical services for which the potential for harm far outweighs the potential for benefit, such as spinal fusions for back pain, Pap smears for elderly women, and meniscus removal for degenerative knee joints. Researchers found that health systems that were primarily investor-owned performed more low-value care. In contrast, the systems that had the lowest levels of low-value care were those with a higher concentration of primary care physicians, a medical teaching program, and those that provide more uncompensated care. Segal et al., “Factors Associated with Overuse of Health Care Within U.S. Health Systems: A Cross-sectional Analysis of Medicare Beneficiaries From 2016 to 2018,” JAMA Health Forum, 1/14/2022
Nonprofit hospitals got big tax breaks with little charity care: Out of the 275 nonprofit hospital systems, 227 had “fair share deficits,” meaning they spent less on charity care and community investments than they received in tax breaks. Total “fair share deficits” amounted to $18.4 billion in 2019. The top five offenders were Providence Saint Joseph Health ($705 million more in tax breaks than it spent on charity care), Trinity Health ($671 million), Mass General Brigham ($625 million), Cleveland Clinic Health System ($611 million), and UPMC ($601 million). “Fair Share Spending: How much are hospitals giving back to their communities?,” Lown Institute, 4/12/2022
Majority of physicians now corporate employed: Physician practice acquisitions increased during the COVID-19 pandemic, and now nearly three out of four (74%) physicians are employed by hospitals, health systems, and other corporate entities such as private equity firms and health insurers. Nationally, hospitals and other corporations acquired 36,200 additional physician practices between 2019 and 2021, leading to a 38% increase in the percentage of corporate-owned practices. During that time, more than 108,000 physicians became employees of hospitals or other corporate entities (with 58,000 in hospitals and 50,500 in other corporate entities), a 19% increase. More than 75% of those employees (83,000) made the shift after the start of the pandemic. “COVID-19’s Impact on Acquisitions of Physician Practices and Physician Employment 2019-2021,” Avalere Health, April 2022
Another year, another drug price hike: In early 2022, pharmaceutical companies raised wholesale prices by a median of 4.9% on more than 450 prescription medicines. United Therapeutics increased the price of its childhood cancer medication, Unituxin, by 9.9%, following a 9.9% increase in 2021 (it now retails for $14,349 per vial). Recordati raised the price of Neoprofen, a drug that treats premature infants at risk of a congenital heart defect, by 10% in 2020, 2021, and 2022 (it now retails for nearly $3,000). Leadiant hiked the price of a 50-year old cancer drug, Matulane, by more than 15% (it now retails for $11,969). Vertex raised the price of Trikafta, a cystic fibrosis medication that has no competitors and already has a list price of more than $311,000 for an annual supply, by 4.9% in 2022. Pfizer raised prices for roughly 100 drugs, including a 16.8% price hike for its injectable hydrocortisone product and a 6.9% increase on breast cancer drug Ibrance. Silverman, “Drug makers ring in the new year with 5% price hikes on hundreds of medicines,” 1/03/2022; “Brand Drug List Price Change Box Score,” 46Brooklyn Research, 1/05/2022
Drug prices outpace inflation: Half of all drugs covered by Medicare Part D (50% of 3,343 drugs) and nearly half of all Part B covered drugs administered by a physician (48% of 568 drugs) had price increases greater than inflation between July 2019 and July 2020. Among those drugs, one-third (668 drugs) had price increases of 7.5% or more. Among the most expensive price hikes were Eliquis, a blood thinner used by 2.6 million beneficiaries, with a 5.9% price increase; Revlimid, a treatment for multiple myeloma used by 44,000 beneficiaries, with a 6.5% increase; and Xarelto, a blood thinner used by 1.2 million beneficiaries, with a 4.1% price increase. Cubanski and Neuman, “Prices Increased Faster Than Inflation for Half of all Drugs Covered by Medicare in 2020,” Kaiser Family Foundation, 2/25/2022
“Response To: Cost-Sharing: Implications of a Well-Intended Benefits Strategy,” by Shannon M Rotolo, PharmD, BCPS, Journal of Managed Care and Specialty Pharmacy, May 2022. “United States could move toward a single-payer system with no premiums, no deductibles, and no copayments. Pharmacists in patient-facing roles would no longer need to provide support or coaching on health insurance literacy and no longer need to help patients choosing a plan that is “best” for them but potentially still inadequate. All patients would have consistent and transparent coverage from birth to death, regardless of their socioeconomic status.”
“Response to: A Potential Path to Universal Coverage With Medicare Advantage for All,” by Adam Gaffney, MD, MPH; David U. Himmelstein, MD; Steffie Woolhandler, MD, MPH. “Patients want good health care coverage and unrestricted choice of physicians and hospitals, not, as this Viewpoint suggests, choice of which insurance plan processes the bill. ‘MA for All’ would perpetuate the upward spiral of health care spending, divert more medical resources to insurers, and restrict choice. Far from looking to MA as a model for reform, we should question whether it should play any role at all.”
“Healthcare and Racial Justice: Systemic Change Is Needed for a More Equitable Health System,” by The Campaign for NY Health and the Black, Puerto Rican, Hispanic and Asian Legislative Caucus, 5/04/2022. Using New York State level data, the report found that Black Americans are 10% less likely than their white counterparts to hold employer-sponsored health coverage, and are also more likely to hold medical debt and to die from pregnancy-related causes. Black and Latinx Americans are less likely than white Americans to have jobs that permit remote work; and more likely to live in communities experiencing health provider shortages.
“Universal health coverage as hegemonic health policy in low- and middle-income countries: A mixed-methods analysis,” by Daniel Smithers, MD and Howard Waitzkin, MD, Social Science and Medicine, June 2022. Globally, the concept of “universal health coverage” (UHC) usually calls for public spending to buy health insurance from private corporations for those without insurance, as opposed to “Health care for all” (HCA), which provides the same comprehensive services for an entire population. UHC has become the dominant policy option favored by political and economic elites around the world, but further enhances the wealth and power of private corporations, without providing comprehensive services to all people.
“Universal healthcare as pandemic preparedness: The lives and costs that could have been saved during the COVID-19 pandemic,” by Alison P. Galvani, Alyssa S. Parpia, Abhishek Pandey, Pratha Sah, Kenneth Colón, Gerald Friedman, Travis Campbell, James G. Kahn, Burton H. Singer, and Meagan C. Fitzpatrick, PNAS, 6/13/2022. The fragmented and inefficient healthcare system in the U.S. leads to many preventable deaths and unnecessary costs every year, especially during a pandemic. Researchers estimate that a single-payer universal health care system would have saved 212,000 lives in 2020 alone, and also calculated that$105.6 billion of medical expenses associated with COVID-19 hospitalization could have been averted by a Medicare for All system.
“Medical Documentation Burden Among U.S. Office-Based Physicians in 2019: A National Study,” by Adam Gaffney, MD, MPH, Stephanie Woolhandler, MD, MPH, Christopher Cai, MD, David Bor, MD, Jessica Himmelstein, MD, Danny McCormick, MD, MPH, David U. Himmelstein, MD, JAMA Network, 3/28/2022. In this cross-sectional study, U.S. physicians spent a mean of 1.77 hours daily completing documentation outside office hours in 2019, and an estimated 125 million hours total documenting outside office hours. Nearly 57% percent of physicians said time spent documenting reduces the time they can spend with their patients. Relative to EHR users in other nations, U.S. physicians spend more time documenting in the EHR, with outpatient notes approximately times longer.
“COVID-19 Testing and Incidence Among Uninsured and Insured Individuals in 2020: a National Study,” by Adam Gaffney, MD, MPH, Steffie Woolhandler, MD MPH, and David U. Himmelstein, MD, Journal of General Internal Medicine, 2/09/2022. In 2020, uninsured adults (including many with chronic diseases) were less likely than the insured to have been tested for COVID-19 despite having higher rates of positive test results. Nearly one-third (32.8%) of the insured were tested compared to 26.2% of the uninsured. Among those tested, the positivity rate was almost double among uninsured (21.7%) relative to insured (11.1%) individuals.
“Health Care Debt In The U.S.: The Broad Consequences Of Medical And Dental Bills,” by Lunna Lopes, Audrey Kearney, Alex Montero, Liz Hamel, and Mollyann Brodie, Kaiser Family Foundation, 6/16/2022. Substantial shares of adults carry debt from medical and dental bills that they have paid off by taking on other forms of debt, including credit cards, personal bank loans, or loans from family and friends. The KFF Health Care Debt Survey finds that four in ten adults have some form of health care debt. Yet the likelihood of having health care debt is not evenly distributed. Uninsured adults, women, Black and Hispanic adults, parents, and those with lower incomes are especially likely to say they have health care-related debt.
In Arizona, PNHP members Drs. Michael Hamant and Eve Shapiro successfully introduced and passed a resolution at the annual meeting of the Arizona Medical Association demanding that the Dept. of Health and Human Services and President Biden administration immediately end Medicare Direct Contracting and REACH. Drs. Hamant and Shapiro explained to their colleagues that since most physicians are now employed by large groups or health systems, they may find themselves practicing in DCEs without their knowledge or consent. To get involved in Arizona, contact Dr. Shapiro at evecshapiro@gmail.com.
In Colorado, PNHP leaders helped to organize and host the One Payer States Meeting on May 21. The conference was co-sponsored by PNHP-CO, the Colorado Foundation for Universal Health Care and Health Care for All Colorado. To get involved in Colorado, contact Dr. Rick Bieser at rgbieser@gmail.com.

In Georgia, a mix of new and experienced PNHP members — ranging from medical students to retirees — gathered in April to discuss single payer and celebrate the founding of three new Georgia Students for a National Health Program (SNaHP) chapters at Emory, Morehouse, and the Medical College of Georgia. In May, PNHP-GA assembled a Steering Committee to coordinate statewide education and organizing campaigns; the Committee’s first task is to work with allies like National Nurses United (NNU) to request that Georgia Senators Warnock and Ossoff co-sponsor the Senate single-payer bill. To get involved in Georgia, contact Dr. Elizabeth McCord at eomccord@gmail.com.
In Illinois, Dr. Pam Gronemeyer of southern Illinois worked with allies in Missouri to pass a Medicare-for-All resolution in the St. Louis City Council (see Missouri report). PNHP-IL members worked with other local health advocates to protest the closing of CVS pharmacies in underserved neighborhoods; co-chair Dr. Anne Scheetz spoke at the coalition’s May 27 press conference in Chicago. To get involved in Illinois, contact Dr. Monica Maalouf at mmaalouf88@gmail.com.

In Kentucky, PNHP members have given presentations about Medicare for All to several candidates running for Congress, including one Republican. PNHP’ers have also been active in the campaign to protect Medicare from Direct Contracting and REACH by publishing opinion pieces in local news outlets and winning endorsements for the campaign from a number of Kentucky organizations, including the Kentucky AFL-CIO. To get involved in Kentucky, contact Kay Tillow at nursenpo@aol.com or Dr. Garrett Adams at kyhealthcare@aol.com.
PNHP’s Missouri chapter spent much of the past year organizing a diverse coalition in support of a Medicare-for-All resolution in the St. Louis City Council. On March 4, Drs. Nat Murdock, Monique Williams, and Ed Weisbart presented the resolution to the City Council’s Black Caucus. Then on May 12, Dr. Weisbart, Angela Brown (CEO of the St. Louis Regional Health Commission), and patient advocate Chris Wilcox presented the resolution to the City’s Health and Human Services Committee, with a unanimous vote to support the resolution by both the committee and the full Board of Aldermen. To get involved in Missouri, contact Dr. Weisbart at pnhpMO@gmail.com.

In New Jersey, PNHP-NY Metro board member Dr. Leonard Rodberg made a presentation about Medicare Direct Contracting/REACH to the NJ Universal Healthcare Coalition meeting on April 23. The coalition is working to pass resolutions in the Medical Society of New Jersey, including a resolution supporting Medicare for All and a resolution opposing Direct Contracting/REACH. To get involved in New Jersey, contact Dr. Lloyd Alterman at lloydalterman52@gmail.com.
In New York, PNHP’s New York-Metro chapter continues its monthly education forums; recent topics include pharmaceutical pricing, and abortion and reproductive health care in a post-Roe environment. NY-Metro held its annual Lobby Day on May 3, where advocates met with 13 state legislators to ask their support for the single-payer NY Health Act as well as the End Medical Debt Act, a new bill that would prohibit hospitals and insurance companies from placing liens on patients’ property or garnishing wages to pay medical debt. The chapter is also helping New York City public worker retirees fight the forced transition from Traditional Medicare to Medicare Advantage by urging NYC City Council members to sign a letter to the mayor asking him to keep the TM plan. On May 14, NY-Metro chapter members joined the “Bans Off Our Bodies” abortion rights march across the Brooklyn Bridge; several members provided medical support to marchers. This summer, the chapter is implementing a new communications strategy which includes publishing several letters and op-eds in local news outlets, as well as a social media campaign called #MedStoryMondays, where medical workers share their stories of how the current system has failed them or their patients. To get involved in New York, contact Mandy Strenz at mandy@pnhpnymetro.org.

Health Care Justice – NORTH CAROLINA in Charlotte held its annual membership meeting on March 31 with 70 health care advocates in attendance, including several elected officials. The group focused on the dangers of profiteering in Medicare through Direct Contracting/REACH. Chapter members participated in the May 14 “Bans Off our Bodies” rally for abortion care. Thanks to organizing by the North Carolina Medicare For All Coalition (a statewide coalition of 45 organizations), the North Carolina Democratic Party officially added single-payer Medicare for All to its platform at the party’s meeting on June 17. To get involved in Health Care Justice-NC, contact Dr. Jessica Schorr Saxe at jessica.schorr.saxe@gmail.com.
Health Care for All NC Raleigh members have been active in the Poor People’s Campaign, sending Medicare for All postcards to elected officials and sending a delegation of health care workers to the PPC Moral March in Washington on June 18. Chapter leaders have also been engaged in speaking events: Drs. Jonathan Kotch and Howard Eisenson spoke about single payer to faculty and trainees at Duke Medicine, and Jonathan Michels spoke to students at the Wake Early College of Health and Sciences who are now forming their own SNaHP chapter. To get involved in Health Care for All NC in Raleigh, contact Jonathan Michels at jonscottmichels@gmail.com.
Members of Healthcare For All – Western North Carolina in Asheville met with U.S. Senate candidate Cheri Beasley to urge her support for Medicare for All. Chapter leaders also hosted presentations on Medicare privatization through Direct Contracting and REACH at two large retirement communities in Asheville. To get involved in HCFAWNC, contact Terry Hash at theresamhash@gmail.com.
In Pennsylvania, PNHP’s Eastern PA chapter leader Dr. Walter Tsou gave a Grand Rounds on COVID-19, institutional racism, and health reform at Penn Family Medicine. Health Care for All Philadelphia participated in the annual “Mt. Airy Day” event where they explained their opposition to Medicare Direct Contracting and REACH to elected officials in attendance. To get involved in Pennsylvania, contact Dr. Tsou at macman2@aol.com.

PNHP Vermont has given several presentations about the privatization of Traditional Medicare through Direct Contracting and REACH; several of these presentations have been recorded and broadcast on local access television stations. The PNHP Northern New England Summer internship just wrapped up with 13 medical students from Kentucky, Missouri, Arizona, Texas, Louisiana, Florida, New Jersey, New York, and Michigan. To get involved in Vermont, contact Dr. Betty Keller at bjkellermd@gmail.com or Ted Cody tscody@vermontel.net.
PNHP Washington has been very active in the campaign to stop Medicare privatization. Several of the chapter’s monthly educational forums have been focused on this topic, including a talk from Donald Cohen, author of “The Privatization of Everything,” as well as several training sessions on Direct Contracting and REACH. PNHP-WA worked closely with Puget Sound Advocates for Retirement Action, Health Care is a Human Right WA, and other allies to successfully pass an anti-DC/REACH resolution in the Seattle City Council. They also met with staff for U.S. Rep. Suzan DelBene, Sen. Maria Cantwell, and Sen. Patty Murray, urging them to fight back against DC/REACH. To get involved in Washington, contact pnhp.washington@gmail.com.
In West Virginia, PNHP and SNaHP members meet monthly to plan organizing tasks and discuss health care policy topics. Chapter members have given presentations about Direct Contracting/REACH to senior groups and will continue outreach to other health reform groups. Chapter leaders are actively recruiting new members at the state’s FQHCs with the goal of having at least one active member in each of the state’s 55 counties. To get involved in West Virginia, contact Dr. Dan Doyle at pnhp.wv@gmail.com.
“Seniors’ Medicare Benefits Are Being Privatized Without Consent,” The Lever, 4/11/2022, featuring Dr. Ed Weisbart and Kip Sullivan
“Secret Trump Program To Privatize Medicare Needs to End,” The Thom Hartmann Program, 5/02/2022, featuring Dr. Susan Rogers
“New Stealth Attack on Medicare Opens Door to Privatization,” Rising Up with Sonali, 5/24/2022, featuring Dr. Ana Malinow
The Annual Meeting and related events were held at the Boston Park Plaza, located at 50 Park Plaza at Arlington Street, Boston, MA 02116.
PNHP’s Board of Directors and medical experts have established Covid safety protocols for the Annual Meeting that include a surgical facemask requirement while not actively eating or drinking; proof of vaccine within the last 5 months (physical card, photo, or electronic record is fine); and proof of a negative Covid test (time-stamped photo of a rapid antigen test is fine) taken within 24 hours of the Summit.
See below for final scheduled; conference speakers included:

SNaHP Summit: Friday, Nov. 4, 8:00 a.m. to 12:00 p.m. Final schedule HERE
Leadership Training: Friday, Nov. 4, 1:00 to 5:00 p.m.; small group meetings 5:00 to 6:30 p.m.; dinner 6:30 to 8:00 p.m. Final schedule HERE
Annual Meeting: Saturday, Nov. 5, 9:00 a.m. to 5:00 p.m.; small group meetings 5:00 to 7:00 p.m.; dinner (with programming) 7:00 to 9:00 p.m. Final schedule HERE
To request a meeting for a Member Interest Group (MIG), please contact deputy director Matthew Petty at matt@pnhp.org
This conference was not livestreamed in its entirety, but recordings of select sessions are available at pnhp.org/boston22.
Scholarships were provided to 74 students and residents, helping to cover the cost of travel, lodging, and registration.
PNHP members and the public can support PNHP’s student outreach programs by making a GIFT to the Nicholas Skala Student Fund.
Public health workers at the APHA Annual Meeting participated in a “Rally for Public Health Reproductive Justice” on Sunday, Nov. 6 at 1:00 p.m. The rally was held outside the Boston Convention and Exhibition Center entrance on Summer St.
This demonstration against the recent Dobbs v. Jackson Women’s Health Organization decision and continuing attacks on reproductive justice was endorsed by multiple APHA sections and caucuses (Maternal Child Health, Black, Women’s, Peace, LGBT, Socialist, and others); Massachusetts Planned Parenthood; Medical Students for Reproductive Choice; and Physicians for National Health Program.
The 2022 Health Activist Dinner at APHA was held Sunday, Nov. 6 from 6:00 to 9:00 p.m. PNHP was a co-sponsor of this event.
This year’s honorees included Michelle Morse, M.D., M.P.H. (Paul Cornely Award); Riyadh Lafta, M.B.Ch.B., Ph.D. (Edward Barsky Award); and Rep. Ayanna Pressley (Paul Wellstone Award). Ted Brown, Ph.D., delivered a keynote address on “A Heroic History of the U.S. Health Left.”
Click HERE to view photos from our previous in-person Annual Meeting (2019 in Philadelphia).
Click HERE for materials from our (fall) 2021 virtual Annual Meeting, HERE for materials from our (spring) 2021 virtual Leadership Training, and HERE for materials from our (fall) 2020 virtual Annual Meeting.
Attending the 2022 PNHP Annual Meeting is entirely voluntary and requires attendees to abide by any applicable rules of conduct or local or state laws that may be announced at any time. Attendees acknowledge the highly contagious and evolving nature of Covid-19 and voluntarily assume the risk of exposure to or infection with the virus by attending the Meeting and understand that such exposure or infection may result in personal injury, illness, disability, and/or death. Attendees release and agree not to sue any persons or entities responsible for coordinating or organizing the PNHP Annual Meeting in the event that they contract Covid-19. Attendees agree to comply with all Covid-related procedures that may be implemented at the Meeting, including mask-wearing.
Click the links below to jump to different sections of the newsletter. To view a PDF version of the shorter print edition of the newsletter, click HERE.
If you wish to support PNHP’s outreach and education efforts with a financial contribution, click HERE.
If you have feedback about the newsletter, email info@pnhp.org.
PNHP leads campaign against Direct Contracting
On February 24, the Centers for Medicare and Medicaid Services (CMS) made a stunning announcement: The agency was terminating the controversial Medicare Direct Contracting (DC) program four years ahead of schedule, and “rebranding” the program into a new pilot called ACO REACH.
The announcement came less than three months after a delegation of PNHP physicians and medical students launched a campaign against DC with a press conference at the headquarters of CMS’ parent agency, the Department of Health and Human Services. There, PNHP leaders delivered a stack of petitions to HHS Secretary Xavier Becerra, demanding that he immediately end DC and keep Medicare public for future generations.

What is Direct Contracting?
Medicare DC is a pilot program developed during the Trump Administration that would change the way that Traditional Medicare pays for care. Instead of paying providers directly, Medicare pays third-party middlemen called Direct Contracting Entities (DCEs) to “coordinate” beneficiaries’ care. DCEs are allowed to keep up to 40% of these payments as profit and overhead, a dangerous incentive to restrict patient care.
Medicare beneficiaries are automatically enrolled into DCEs without their understanding or consent, and once enrolled, must change primary care providers to opt out. Virtually any type of company can apply to be a DCE, including those owned by commercial insurers, private equity investors, and for-profit dialysis centers. Even though DC is technically a “pilot program,” CMS can scale the program up to all of Traditional Medicare without the approval of Congress.
Campaign wins support in Washington
When the physicians traveled to Washington in November, most members of Congress had never even heard of DC. PNHP members and allies quickly met with their representatives and soon won the support of several influential leaders, such as Medicare for All lead sponsor Rep. Pramila Jayapal. In December, Rep. Jayapal and PNHP president Dr. Susan Rogers published an op-ed in The Hill, “The biggest threat to Medicare you’ve never even heard of,” which was shared thousands of times.
In January, Rep. Jayapal and more than 50 Congressional colleagues sent a letter to HHS Sec. Becerra demanding he immediately end the DC program. The campaign then caught the attention of Sen. Elizabeth Warren, who invited Dr. Rogers to speak about Medicare privatization at a Senate Finance committee hearing on Feb. 2.

In the meantime, PNHP members published op-eds and letters-to-the-editor, and campaign leaders gave dozens of talks and media interviews. By late February, the campaign generated 80 news articles and nearly 50 opinion pieces. In a matter of months, “Direct Contracting” was synonymous with corruption, profiteering, and privatization.
CMS response to the campaign was to “rebrand” DC into the REACH program. Unfortunately, REACH carries over all the most dangerous aspects of DC, and would continue to allow Wall Street middlemen to profit at the expense of Medicare and its beneficiaries.
PNHP immediately responded to CMS’ rebranding with a press statement and video slamming REACH. Less than two weeks later, the campaign sent a letter to CMS and HHS from more than 250 organizations representing health providers, seniors, disabled adults, unions and community groups, demanding an end to REACH and Medicare privatization.
The next phase of the fight against privatization
“The Direct Contracting campaign taught us two important lessons,” said PNHP president Dr. Susan Rogers. “First, health justice advocates have incredible power when we educate, organize, and speak out. Second, when Wall Street profiteers get their hands in public programs like Medicare, it’s not easy to get them out.” Dr. Rogers noted that PNHP and allies would have to redouble their efforts to end REACH, while also fighting against the growth of Medicare Advantage, the version of Medicare run by commercial insurers for profit. “If we want Medicare for All tomorrow, we must fight to keep Medicare public today,” said Dr. Rogers.

Single-payer advocates across the country have been signing up additional Congressional co-sponsors on H.R. 1976, the Medicare for All Act of 2021. When the bill was introduced by Rep. Pramila Jayapal last March, it had 112 co-sponsors. In the weeks after the introduction, activists quickly signed up another three sponsors (Reps. Mike Quigley of Illinois, Kweisi Mfume of Maryland, and Betty McCollum of Minnesota). Over the summer, two more members of Congress (Reps. Zoe Lofgren of California and Melanie Ann Stansbury of New Mexico) signed on. And in just the last few months, the movement signed up another four co-sponsors (Reps. John Garamendi of California, Shontel Brown of Ohio, Donald Norcross of New Jersey, and Sheila Cherfilus-McCormick of Florida), bringing the total sponsors to 121, the highest number of sponsors ever on a single-payer bill. In every case, the representatives only signed on after sustained pressure from activists, including phone calls, letters, social media “storms” and public actions. For more information about Medicare for All legislation, visit pnhp.org/HouseBill.
Two more state medical associations have recently joined PNHP’s Medical Society Resolutions campaign. In September, PNHP members passed a resolution in the Washington State Medical Association expressing support for “universal access to comprehensive, affordable, high-quality health care … including a publicly-funded national health care program.” The resolution passed with 93% of the vote.
PNHP’s Granite State chapter launched their resolution campaign by sending a survey to the New Hampshire Medical Society. They found that 82% of primary care doctors and 66% of specialists support Medicare for All, and similar percentages would support single payer as the official position of the NHMS. Confident in their success, activists submitted a single-payer resolution to the NHMS Council, which passed by an overwhelming majority in March.
Washington State and New Hampshire join Vermont and Hawaii in passing single-payer resolutions in their state medical societies. To get involved in PNHP’s medical society resolutions campaign, visit medicalsocietyresolutions.org or contact organizer@pnhp.org.
The Congressional Budget Office, the federal agency that provides Congress with economic analysis of proposed laws and policies, just released a powerful report on the potential impact of Medicare for All on the U.S. economy. The CBO concluded that single payer would have several positive impacts on workers and households, as well as the national economy:
To learn more about this study and the economic impact of Medicare for All, visit pnhp.org/PayingForIt.

Americans delay or skip treatment because of cost. By late 2021, nearly one-third (30%) of Americans reported not seeking treatment for a health problem in the prior three months due to cost, a percentage that tripled since March. One-fifth of adults (21%) reported a member of their household had a health problem worsen after postponing care because of cost. The rate of Americans borrowing money to pay for needed care rose from 7% in December 2020 to 11% in October 2021. An estimated 12.7 million Americans report knowing a friend or family member who died this past year after not receiving treatment because of cost, and Black Americans (8%) are twice as likely to know someone who died as whites (4%). In 2021, many Americans cut back on other necessities to pay for care, including clothing (26%), food (13%) and utilities (8%). “2021 healthcare in America report,” West Health-Gallup, 12/14/2021
Americans routinely delay or skip needed care. In the past two years, 33% of Americans skipped dental care, 25% delayed a doctor visit or procedure, 24% completely skipped a visit or procedure, 18% avoided going to the hospital or ER, and 15% experienced pain because they could not afford medical care. People with ACA Marketplace plans (78%) were much more likely to report delaying or skipping care due to costs than those with any other type of coverage. “Healthcare affordability: Majority of adults support significant changes to the health system,” Robert Wood Johnson Foundation, August, 2021
18 million Americans can’t afford needed medications. Seven percent of U.S. adults (18 million) were unable to pay for at least one doctor-prescribed medication for their household in early 2021, with higher rates among low-income households. In households earning less than $24,000, almost 20% were unable to pay for medications; for those earning less than $48,000, 18% report skipping pills. About 1 in 10 adults say they’ve skipped a pill in the prior year to save money. Witters, In U.S., an Estimated 18 Million Can’t Pay for Needed Drugs, Gallup, 9/21/2021
Nearly one in ten Americans hold medical debt. Roughly 23 million people, or 9% of American adults, owe medical debt, including 11 million who owe more than $2,000 and 3 million who owe more than $10,000. Americans’ collective medical debt totaled at least $195 billion in 2019, with people in middle age (35-64) more likely than other adults to report medical debt. Larger shares of people in poor health (21%) and living with a disability (15%) report medical debt, as well as a larger share of Black adults (16%) compared to White (9%), Hispanic (9%), and Asian American (4%) adults. Rae et al. “The burden of medical debt in the United States,” Kaiser Family Foundation, 3/10/2022
Majority of Americans’ debt is medical. In a new survey, roughly 20% of U.S. households report having medical debt, and medical collections tradelines appear on 43 million credit reports. As of mid- 2021, 58% of bills that are in collections and on people’s credit records are medical bills. Black Americans are twice as likely (28%) to have past-due medical debt compared to white Americans (17%), and debt is more common in regions that did not expand Medicaid. Medical bills on credit reports can result in reduced access to credit, increased risk of bankruptcy, avoidance of medical care, and difficulty securing employment, even when the bill itself is inaccurate or erroneous. “Medical Debt Burden in the United States,” Consumer Financial Protection Bureau, February 2022
Medical debt disproportionately impacts the poor. Nearly 80% of medical debt is held by households with zero or negative net worth. Only 9% of medical debt is held by households with between $1 and $104,000 in net worth, and surprisingly, 13% of medical debt is held by households with more than $104,000. Among those with insurance coverage, 26% percent of Black households hold medical debt compared to 16% of non-Black households. Among the uninsured, 35% percent of Black households hold medical debt compared to 26% of non-Black households. Perry et al., “The racial implications of medical debt: How moving toward universal health care and other reforms can address them,” Brookings Institution, 10/5/2021
High out-of-pocket costs drive up underinsurance in children. Underinsurance for children (lack of continuous and adequate insurance) rose from 31% in 2016 to 34% in 2019 — an additional 2.4 million children — driven primarily by unaffordable out-of-pocket medical expenses. The recent growth of children’s underinsurance was driven by those in white and mulitracial households, those considered middle-class (incomes above 200% of poverty), and those with private health insurance. Yu et al., “Underinsurance among children in the United States,” Pediatrics, January 2022
Despite Medicare, American seniors face financial barriers to care. One in five U.S. seniors (20%) pay more than $2,000 out of pocket for health care services, while most other wealthy nations average 5% or less. In the U.S., 8% of seniors delayed or avoided care in the past year, and 9% skipped medications, compared to 2% or less in countries like Germany and Sweden. Jacobson, et al., “When costs are a barrier to getting health care: Reports from older adults in the U.S. and other high-income countries,” Commonwealth Fund, 10/01/2021
U.S. life expectancy plummets, trails other higher-income nations. U.S. life expectancy dropped by 2.27 years in men and 1.61 years in women in 2020, the largest drop of any other middle or high-income country except for Russia. The U.S. drop in life expectancy was driven largely by the deaths of young people. In contrast, in several countries with strong public health and universal coverage — New Zealand, Taiwan, Iceland, South Korea, Norway and Denmark — life expectancy either increased or remained the same. Islam et al., “Effects of Covid-19 pandemic on life expectancy and premature mortality in 2020: Time series analysis in 37 countries,” BMJ, 11/03/2021
Maternal mortality crisis grows. The U.S. maternal mortality rate increased by 18% from 2019 to 2020, from 20.1 deaths to 23.8 deaths per 100,000 live births; the 2020 rate is a 37% increase from 2018. Black people had the highest maternal mortality rate (55.3 deaths/100,000 live births), nearly three times higher than white people. The maternal death rate among Hispanic people rose by 44% from 2019 to 2020. The U.S. maternal mortality rate was more than double that of other developed countries. Hoyert, “Maternal mortality rates in the United States, 2020,” National Center for Health Statistics,” February 2022
Latinx/Hispanic Americans have higher rates of preventable cancer. Due to a lack of access to care, Latinx/Hispanic individuals are more likely to suffer from potentially preventable cancers compared to whites. In 2018, 26% of Latinx/Hispanic individuals were uninsured, compared to 9% of whites. Latinx/Hispanic people are more than twice as likely as white people to develop liver cancer or stomach cancer, and are twice as likely to die from those cancers. Compared to white women, Latinas are 32% more likely to get cervical cancer which is almost entirely preventable through screening and vaccination. Miller et al., “Cancer statistics for the U.S. Hispanic/Latino population, 2021,” CA: A Cancer Journal for Clinicians, 9/21/2021
Inequities plague cancer detection and treatment. While overall risk of cancer death is much lower than it was decades ago, racial and geographic disparities persist. Risk of cancer death is 33% higher for Black people and more than 50% higher in Native Americans and Alaska Natives, compared with white people. Even though Black women have a lower rate of breast cancer incidence, they have a 41% higher death rate compared to white women. Breast cancer mortality is also higher in states like Mississippi that did not expand Medicaid and have high levels of poverty. Seigel et al., “Cancer statistics, 2022,” CA: A Cancer Journal for Clinicians, 1/12/2022
VA care reduces both mortality and health spending. For veterans aged 65 and older, getting emergency care in a Veterans Administration (VA) facility reduced 28-day mortality by 46% and reduced 28-day spending by 21% ($2,598) compared to care in a private facility. Chan et al., “Is there a VA advantage? Evidence from dually eligible veterans, Working Paper 29765,” National Bureau of Economic Research, February 2022
Veterans died at lower rates in 2020 compared to the general population. In 2020, American veterans faced an excess mortality rate of 13% in 2020, while the U.S. overall had an excess mortality rate of 23% in the same time frame, despite veterans having higher health risks due to age and conditions like hypertension, diabetes, and obesity. Veterans researchers cite “consistent access to health care and the rapid expansion of VHA telemedicine during the pandemic,” as the reason. Feyman et al., “County-level impact of the Covid-19 pandemic on excess mortality among U.S. veterans: A population-based study,” The Lancet Regional Health – Americas, 10/30/2021
Medicaid expansion saves lives. From 2014 to 2018, Medicaid expansion was associated with nearly 12 fewer deaths per 100,000 adults annually; expansion may lead to an overall 3.8% decline in adult deaths each year. The drop in mortality most benefited women and Black people who are more likely to live in poverty than men and non-Black people, respectively. The greatest reduction in mortality was from respiratory and cardiovascular conditions, suggesting that the decrease in mortality is primarily from greater access to preventive care, specialist referrals, and medications. Lee et al., “Medicaid expansion and variability in mortality in the USA: A national, observational cohort study,” The Lancet, 12/2/2021
Medicare coverage reduces out-of-pocket health spending. Despite a 5% increase in annual medical expenses after 65, older adults’ out-of-pocket health costs dropped by 27%, and their rate of catastrophic health expenditures decreased by 35%, once they enrolled in Medicare at 65. Medicare coverage also led to a 17% reduction in those who delayed seeking care due to cost. Scott et al., “Changes in out-of-pocket spending and catastrophic health care expenditures associated with Medicare eligibility,” JAMA Health Forum, 9/10/2021
Prior authorization (PA) causes disability and death. In a new survey, nearly all (93%) physicians reported care delays while waiting for insurers to authorize necessary care via PA, and 82% said patients abandoned treatment due to PA struggles with insurers. More than one-third (34%) reported that PA led to a serious adverse event for a patient in their care, such as hospitalization, disability, or even death. “2021 AMA prior authorization (PA) physician survey,” American Medical Association, 2/10/2022
Americans skip or delay care due to insurance hassles. One quarter (25%) of insured, working-age adults have either postponed or skipped necessary care because of administrative obstacles. Nearly three-quarters of patients (73%) reported undertaking tasks like scheduling appointments, obtaining prior authorizations, and resolving problems with bills and insurance premiums. Nearly half of patients who encountered issues with premium payments, and more than one-third of patients who experienced billing or prior authorization problems, delayed or skipped care. Kyle and Frakt, “Patient administrative burden in the U.S. health care system,” Health Services Research, 9/08/2021
Insured families can’t afford cost sharing. In 2019, average out-of-pocket spending limits in commercial insurance plans (for in-network services) were $7,900 for an individual and $15,800 for a family. However, nearly half (45%) of single-person, non-elderly households did not have the liquid assets to cover more than $2,000 in costs, and nearly two-thirds (63%) could not cover more than $6,000. Young et al., “Many households do not have enough money to pay cost-sharing in typical private health plans,” Kaiser Family Foundation, 3/10/2022
The cost of job-based insurance is steadily rising. For the nearly 155 million Americans who get health coverage through their jobs, total average annual premiums are now over $22,200 for families, with workers on average paying $5,969 toward the cost of their coverage, and $7,700 for individuals, with workers paying nearly $1,300 towards the cost. “2021 employer health benefits survey,” Kaiser Family Foundation, 11/10/2021
Employer-plan premiums and deductibles outpace family incomes. Average premiums and deductibles in employer-sponsored health plans climbed to $8,070 in 2020, accounting for 11.6% of the U.S. median household income, up from 9.1% in 2010. Rates were as high as 19% of household income in Mississippi and 18% in New Mexico. Workers with single plans paid about 21% of their premiums; those with family coverage paid 29% of their premiums. Collins et al., “State trends in employer premiums and deductibles, 2010–2020,” The Commonwealth Fund,” 1/12/2022
High insurance costs hurt low-income families hardest. Among those with employer coverage, families below 200% of the poverty line spent an average of 10.4% of their income on premiums and medical care, compared to families above 400% of poverty who spent 3.5% of household income on premiums and medical expenses. Claxton et al., “How affordability of employer coverage varies by family income,” Kaiser Family Foundation, 3/10/2022
ACA Marketplace plans increasingly unaffordable, even for higher earners. For families at 400-600% of poverty ($69,680–$104,520 for a family of two), the premium and deductible for an ACA “Bronze” plan represented 18.3% of income in 2015. By 2019, these costs rose to 26.6% of income. For those aged 55-64 years old, the premium alone is now 18.9% of income. Jacobs and Hill, “ACA marketplaces became less affordable over time for many middle-class families, especially the near-elderly,” Health Affairs, November 2021
Big insurers dominate most metro area markets. Nearly three-fourths (73%) of U.S. metro areas were highly concentrated insurance markets in 2020, up from 71% in 2014. Fourteen states had one health insurer that controlled at least half of their markets. In 91% of metro areas, at least one insurer had a commercial market share of 30% or greater, and in 46% of areas, a single insurer’s share was at least 50%. “Competition in health insurance: A comprehensive study of U.S. markets,” American Medical Association, September 2021
UnitedHealth profits in the pandemic. During the second year of the pandemic, the nation’s largest commercial insurer surpassed revenue and profit projections. UnitedHealth’s 2021 revenue was $288 billion, up 12% from 2020, which is triple its revenue from 2010. UH’s 2021 profit was $17.3 billion, also up 12% from 2020, and quadruple its profits from 2010. While the company is known for insurance, a majority of its revenue ($156 billion) comes from its affiliate Optum, which owns physician practices and specialty pharmacies. “UnitedHealth Group reports 2021 results,” UnitedHealth Group, 1/19/2022
Aetna accused of operating a “shadow network” of Medicaid providers. Commercial insurer Aetna, a CVS Health subsidiary, illegally secured contracts with Pennsylvania’s Medicaid program by misrepresenting the number of pediatric providers in its network in order to discourage care and increase profits, according to a federal whistleblower lawsuit. The whistleblower found that many of the providers assigned to the company’s nearly 100,000 child beneficiaries were either not contracted with Aetna, dead, out of state, or did not treat children. Aetna claimed its lower-than-average screening, diagnostic, and treatment rates were due to parental negligence. Tepper, “Aetna lied about provider network to win Medicaid contracts, suit alleges,” Modern Healthcare,” 9/14/2021
Medicare Advantage (MA) costs taxpayers tens of billions more than Traditional Medicare. Medicare overpaid MA insurers by more than $106 billion from 2010 through 2019, with nearly $34 billion during 2018 and 2019 alone. The overpayments were mostly due to upcoding, or MA plans’ fraudulently exaggerating diagnoses to increase patients’ risk scores. In 2019, MA risk scores were 19% higher compared to Traditional Medicare. Under current coding rules, spending on MA is expected to increase by $600 billion from 2023 through 2031, with as much as two-thirds of the increase in spending going toward profits for insurance companies. Schulte, “Medicare Advantage’s cost to taxpayers has soared in recent years, research finds,” Kaiser Health News, 11/11/2021
Medicare Advantage (MA) insurers collect billions for unverified diagnoses. In 2016, MA plans’ fraudulent “upcoding” — using chart reviews and health risk assessments to increase Medicare risk-adjusted payments — cost the federal government $9.2 billion. The nation’s largest insurer, United Healthcare, generated 40% of its risk-adjusted payments, or $3.7 billion, by listing patient conditions unverified through outside medical claims. The top three “upcoded” conditions were vascular disease; major depressive, bipolar and paranoid disorders; and diabetes with chronic complications. Murrin, “Some Medicare Advantage companies leveraged chart reviews and health risk assessments to disproportionately drive payments,” U.S. Department of Health and Human Services, Office of Inspector General, September 2021
Medicare Advantage (MA) plans cited for not paying for care. Four regional MA plans affiliated with UnitedHealthcare and Anthem have been barred from enrolling new members until 2023 after failing to meet the 85% medical loss ratio threshold for three straight years. The enrollment suspensions affected about 80,000 of UnitedHealth’s 7.5 million MA enrollees. Commins, “Anthem, Unitedhealthcare MA Plans sanctioned for missing MLR threshold,” Health Leaders Media, 9/20/2021
Medicare Advantage (MA) market increasingly concentrated. Six insurers control roughly three-quarters of the MA market: UnitedHealth (7.9 million members), Humana (5.1 million), CVS/Aetna (3.1 million), Anthem (1.9 million), Kaiser Permanente (1.8 million), and Centene (1.4 million). Herman, “The big Medicare Advantage players keep getting bigger,” Axios, 1/19/2022
Private equity flows into health care. Acquisitions by private equity (PE) investors in health care have nearly tripled, from $41.5 billion in 2010 to $119.9 billion in 2019, for a total of approximately $750 billion over the last decade, concentrated in home health, physician practices, and outpatient care. Because PE firms are focused on short-term profits, they tend to prioritize revenue over quality of care and engage in unethical billing practices; they also overburden health care companies with debt, strip their assets, and put them at risk of long-term failure. Experts expect PE investment to increase by 30% to 40% in 2022. Scheffler et al., “Soaring private equity investment in the healthcare sector: Consolidation accelerated, competition undermined, and patients at risk,” American Antitrust Institute and the School of Public Health at UC Berkeley, 5/18/2021
Physician management companies and private equity drive up costs. Compared to hospitals that did not use physician management companies (PMCs) for their outpatient departments and ambulatory surgery centers, hospitals with PMCs charged 16.5% higher prices; hospitals with PMCs backed by private equity increased costs by 25%. LaForgia et al., “Association of physician management companies and private equity investment with commercial health care prices paid to anesthesia practitioners,” JAMA Internal Medicine, 2/28/2022
For-profit hospitals avoid unprofitable services. Government-owned and nonprofit hospitals were 9 percentage points and 6.2 percentage points more likely than comparable for-profit hospitals to offer relatively unprofitable services, like psychiatric care, substance abuse treatment, obstetric care, and hospice. For-profits were 32% more likely to offer a profitable service (such as coronary artery bypass grafting surgery) than an unprofitable service, compared to 27.3% for nonprofits and 22.2% for government-owned facilities. Horwitz and Nichols, “Hospital service offerings still differ substantially by ownership type,” Health Affairs, March 2022
Nonprofit insurance CEOs snagged big raises. Across all U.S. health insurers, CEOs received an average 7.5% raise in 2020 compared with 2019. Although nonprofit insurance CEOs were paid less overall than the heads of for-profit insurers, Blue Cross Blue Shield CEOs were more likely to get a substantial raise in 2020. BCBS of Minnesota CEO Craig Samitt got a 109% raise, to $3.3 million; Hawaii Medical Service Association CEO Mark Mugiishi earned a 89.5% raise, to $1.8 million; and Independence Blue Cross’s now-retired CEO Dan Hilferty received a 73.6% raise, to $9.9 million. Tepper, “BCBS CEOs received bigger raises in 2020 than execs at for-profit insurers, report says,” Modern Healthcare, 10/12/2021
Pharma raises prices, spends lavishly on CEOs and ads. A recent Congressional investigation found that from 2016 to 2020, pharmaceutical companies raised the prices of brand-name drugs by 36%, almost four times the rate of inflation. Despite their claims of needing high prices to fund research, drug firms spent heavily on executive salaries and marketing. The 10 largest pharma companies paid their top executives more than $2.2 billion from 2016 to 2020, including nearly $800 million just to their CEOs. AbbVie, Amgen, Novo Nordisk, and Pfizer spent more than $2.6 billion in direct-to-consumer advertising from 2015 to 2018 on just four drugs. “Drug pricing investigation majority staff report,” U.S. House of Representatives Committee on Oversight and Reform, December 2021
Pharma gifts to doctors lead to higher drug spending. Pharmaceutical company gifts to rheumatologists, through food and beverages or consulting fees, are linked with a higher likelihood of prescribing drugs and higher Medicare spending. For each $100 in food/beverage payments, Medicare reimbursement increased 6% to 44% ($8,000 to $13,000). The increases were particularly high for infliximab and rACTH, where a payment of $100 to a prescriber was associated with increases of approximately $72,000 and $30,000 in Medicare reimbursements, respectively. Duarte-García et al., “Association between payments by pharmaceutical manufacturers and prescribing behavior in rheumatology,” Mayo Clinic Proceedings, 2/01/2022
Americans spend almost double what the rest of the world combined spends on drugs. The 20 highest-selling drugs generated $158 billion of global revenue in 2020, but due to our high drug prices, U.S. sales represented 64% of that total, or $101 billion. For 17 of the 20 top-selling drugs worldwide, pharmaceutical firms made more money from U.S. sales than from sales to all other countries in the rest of the world combined. Drugs with the highest revenue disparities between the U.S. and the rest of the world include medications for HIV, autoimmune disease, MS, and diabetes. Claypool and Rizvi, “United we spend: For 20 top-selling drugs worldwide, big pharma revenue from U.S. sales combined exceeded revenue from the rest of the world,” Public Citizen, 9/30/2021
Spending by health industry lobbyists increased in 2021. The Pharmaceutical Research and Manufacturers of America (PhRMA) spent nearly $30 million on lobbying in 2021 — a 16% increase over 2020. Other big spenders include the American Hospital Association, which spent $20.8 million in 2021 versus $18.9 million in 2020; the American Medical Association spent $18.8 million in 2021 (about the same as 2020); and America’s Health Insurance Plans, which spent $11.3 million in 2021. Cigna led the lobbying push among individual insurers, increasing its spending by 27% to $9.1 million in 2021 compared with $7.2 million in 2020. Wilson, “Health interests pour cash into D.C. lobbying,” Politico, 1/24/2022
Albert et al., “Racism, Chronic Disease and Mental Health: Time to Change Our Racialized System of Second-Class Care,” Healthcare, 9/27/2021. PNHP members Drs. Judy Albert, Claire Cohen, Thomas Brockmeyer, and Ana Malinow describe how the “weathering hypothesis” and adverse childhood experiences set the stage for higher rates of chronic disease, mental health disorders, and maternal mortality seen in Black adults in the U.S. They also illustrate the toll that untreated and overtreated mental health disorders have on Black patients, who have similar rates of mental health disorders as their white counterparts but have fewer outpatient mental health services and higher rates of hospitalizations. They also discuss the impact of Medicaid, which now covers 33% of all Black people in the U.S. but suffers from chronic underfunding and administrative burdens on patients. The physicians propose single-payer Medicare for All as a foundation for addressing structural racism in health.
Greep et al., Physician Burnout: Fix the Doctor or Fix the System?, The American Journal of Medicine, 11/01/2021. Even before COVID-19, burnout affected almost half of American physicians, diminishing the quality of physicians’ lives, and increasing the likelihood of medical errors and physicians leaving practice. Drs. Greep, Woolhandler, and Himmelstein argue that America’s profit-oriented, multiplayer system — including commercial insurers’ practice of restricting care through prior authorizations and narrow networks — exacerbates and even causes burnout. The drive for profit often conflicts with physicians’ obligation to prioritize optimal care, and reduces their clinical autonomy. The authors advocate moving to a single-payer system, eliminating out-of-pocket costs, and removing the profit motive from patient care.
Lee et al., “Medicaid expansion and variability in mortality in the USA: A national, observational cohort study,” The Lancet, 12/2/2021. Despite chronic underfunding and major administrative burdens for patients, Medicaid coverage is proven to save lives. Researchers found that from 2014 to 2018, Medicaid expansion was associated with nearly 12 fewer deaths per 100,000 adults annually; expansion may lead to an overall 3.8% decline in adult deaths each year. The drop in mortality most benefited women and Black people who are more likely to live in poverty than men and non-Black people, respectively. The greatest reduction in mortality was from respiratory and cardiovascular conditions, suggesting that the decrease in mortality is primarily from greater access to preventive care, specialist referrals, and medications.
Gaffney and Podolanczuk, “Inequity and the Interstitium: Pushing Back on Disparities in Fibrosing Lung Disease in the U.S. and Canada,” American Journal of Respiratory and Critical Care, 1/06/2022. Drs. Gaffney and Podolanczuk argue that differential access to quality health care can determine which patients live and die from fibrosing interstitial lung disease (fILD). They point to research showing that among U.S. patients with fILD, patients living in the lowest-income communities, death rates were 51% higher compared to their wealthier counterparts. Moreover, U.S. patients with idiopathic pulmonary fibrosis (IPF) residing in the lowest-income neighborhoods were 64% less likely to have a lung transplant compared to those in higher-income neighborhoods. However, these disparities are not present among similar patients in Canada, pointing to the importance of universal coverage.
Gaffney et al., “Association of Uninsurance and VA Coverage with the Uptake and Equity of COVID-19 Vaccination: January–March 2021,” Journal of General Internal Medicine, 1/11/2022. From January to March 2021, the COVID-19 vaccination rate was 55.3% for those with VA coverage vs. 50.1% for those with non-VA coverage, and 30.4% among the uninsured. VA coverage also increased equity: relative to white adults, VA coverage was associated with markedly higher rates of vaccination among Blacks (13.5 percentage points). Researchers conclude that those who lack coverage are less likely to have an established relationship with a primary care provider, and PCPs have proven to be an important potential source of information on vaccines. They also report that equitable access to VA facilities, greater vaccine supply, and direct outreach efforts such as using mobile vaccination units and air-lifting vaccine teams to remote areas, may have contributed to the more equitable and faster vaccine uptake among VA beneficiaries.
Gaffney et al., “COVID-19 Testing and Incidence Among Uninsured and Insured Individuals in 2020: a National Study,” Journal of General Internal Medicine, 2/09/2022. In 2020, uninsured adults — including many with chronic diseases — were less likely than the insured to have been tested for COVID-19, despite having higher rates of positive test results. Researchers conclude that this discrepancy reflects disparities in access to clinicians between the insured and uninsured. These findings also reflect a larger increase in all-cause mortality among the uninsured relative to the insured in 2020.
Gaffney, et al., “Medical Documentation Burden Among US Office-Based Physicians in 2019: A National Study,” JAMA Internal Medicine, 3/28/2022. Many U.S. physicians report that medical documentation is onerous, takes away time from patients, and increases burnout. A new study aims to quantify the burden of medical documentation on office-based physicians. Researchers found that in 2019, doctors spent about 1.8 hours per day documenting electronic medical records outside of the office, which adds up to 125 million hours spent on documentation work outside of regular business hours.
PNHP California worked to pass AB 1400, the state’s single-payer bill. PNHP members provided policy recommendations to the bill’s sponsor, attended rallies and car caravans, wrote letters and op-eds, and published ads in local newspapers in support of the bill. PNHP members also worked to pass several municipal single-payer resolutions, including the cities of Arcata, Blue Lake, Trinidad, and Eureka; Humboldt County; and the community services Districts of Manila and Willow Creek. California members have been very active in PNHP’s campaign to stop Medicare Direct Contracting, organizing a campaign to send hundreds of postcards and emails to Rep. Nancy Pelosi on Valentine’s Day in support of Traditional Medicare. To get involved in California, contact Dr. Corinne Frugoni at cfrugoni@reninet.com.

In Illinois, as part of the PNHP Medical Society Resolutions Campaign, Dr. Peter Orris submitted a single-payer resolution to the Illinois State Medical Society in December; the chapter will organize to pass the resolution in the coming months. Dr. Pam Gronemeyer is collecting signatures for a Medicare for All ballot proposition in downstate Edwardsville. In November, Dr. Duane Dowell spoke at a “Bans Off Our Bodies” rally for abortion rights, which was endorsed by PNHP-Illinois. To get involved in Illinois, contact Dr. Anne Scheetz at annescheetz@gmail.com.
In Kentucky, PNHP members and allies hosted a rally against Medicare Direct Contracting in front of the Humana Insurance building in downtown Louisville (Humana owns a Medicare Advantage business as well as a Direct Contracting Entity). The event’s theme was, “How the Grinch Stole Medicare,” and included a visit by the Grinch himself reading an original Dr. Seuss-style poem about the threat of privatization. To get involved in Kentucky, contact Kay Tillow at nursenpo@aol.com.

PNHP’s Minnesota chapter held their annual meeting in January with nearly 100 members in attendance. The group discussed legislative plans, organizing strategies, and the summer internship, and enjoyed a keynote address from Dr. Rachel Madley, a former SNaHP leader who now works as legislative assistant to Rep. Pramila Jayapal. PNHP and Health Care for All MN braved sub-zero temperatures for a rally at the State Capitol on January 26 to celebrate the formation of the Minnesota Health Plan Caucus, a group of legislators who support single-payer bills in the state House and Senate. In February, the chapter hosted the “Meet the Minnesota Health Plan Caucus” education event, a panel discussion of the Minnesota Health Plan with lead author Sen. John Marty and other caucus leaders to discuss the features of the bill, how to organize to support it, and the anticipated timeline to get it passed. To get involved in Minnesota, contact pnhpminnesota@gmail.com.

In New Hampshire, PNHP’s Granite State chapter sent a survey to the New Hampshire Medical Society, gauging their support for single payer. They found that 82% of primary care doctors and 66% of specialists support Medicare for All, and similar percentages said they would support making single payer the official position of the NHMS. In response, activists submitted a single-payer resolution to the NHMS Council, which passed in March with an overwhelming majority. PNHP activists and allies also worked to pass a Medicare for All resolution in the City Council of Keene, making it the sixth NH town to pass a MFA resolution in the last two years. To get involved in New Hampshire, contact Dr. Donald Kollisch at donald.o.kollisch@dartmouth.edu.

In New York, PNHP-NY Metro has built working groups to implement chapter advocacy projects including Medicare privatization, the single-payer New York Health Act, and recruitment of doctors and other health workers into the movement. The chapter has worked hard to block the city’s scheme to move 250,000 municipal retirees into a Medicare Advantage plan by holding educational webinars; writing and distributing detailed analyses of the financial impact on retirees and health inequities; submitting an affidavit supporting a lawsuit; and supporting a retiree resistance group that held several rallies against the mayor’s decision. In March, a judge rejected the city’s plan, although the ruling is being appealed. NY-Metro continues to hold online monthly forums on topics like Direct Contracting and Medicare privatization, and how to fight back against high prescription drug prices. To get involved in NY-Metro, please contact Executive Director Bob Lederer at bob@pnhpnymetro.org.
In North Carolina, Health Care Justice NC members helped persuade Rep. Alma Adams to sign the Congressional sign-on letter against Medicare Direct Contracting, which was released in January. The chapter has developed a strong relationship with Rep. Adams and her staff over the years. To get involved in Health Care Justice NC, contact Dr. Jessica Schorr Saxe at jessica.schorr.saxe@gmail.com.
Members of Health Care for All Western North Carolina (HCFA-WNC) in Asheville have been active in the campaign against Medicare Direct Contracting, holding a watch party for PNHP President Dr. Susan Rogers’ testimony at the Senate Finance Committee in February, and gathering signatures for the Direct Contracting Petition and organizational sign-on letter. To get involved in HCFA-WNC, contact Terry Hash at theresamhash@gmail.com.
In Ohio, PNHP members and allies organized a car caravan in Columbus, urging Rep. Joyce Beatty to sign onto H.R. 1976, The Medicare for All Act. Activists amplified the pressure with an ad targeting Rep. Beatty in the Columbus Dispatch. Allies organized similar events on the same day in three other cities in Ohio, targeting members of Congress who had not signed on to H.R. 1976. To get involved in Ohio, contact Dr. Jim Binder at jamesbinder3@gmail.com.

In Oregon, the PNHP chapter is holding weekly “Conversations with Candidates” health policy forums for candidates running for governor and Congress. Sixteen candidates have agreed to be interviewed, and each forum attracts more than 50 audience participants. Chapter members have also been active in rallies across the state for Medicare for All. To get involved in Oregon, contact Dr. Peter Mahr at peter.n.mahr@gmail.com.
In Vermont, PNHP members collaborated with multiple health care activists to send a letter to CMS and to the Vermont Attorney General regarding OneCare, a state program similar to Medicare Direct Contracting/REACH. Chapter leaders also met with several candidates to discuss their positions on Medicare for All, including staff representing U.S. Senator Patrick Leahy and Rep. Peter Welch, as well as Lieutenant Governor Molly Gray, and State Senators Becca Balint and Kesha Ram-Hinsdale. The PNHP New England coalition is once again hosting a summer internship for medical students. This spring, they interviewed and selected students for the 2022 session, which will include students from New Jersey, Arizona, Louisiana, Florida, and New York. To get involved in Vermont, contact Dr. Betty Keller at bjkellermd@gmail.com.
In Washington, chapter members met with key staff of their Congressional representatives, including Sen. Patty Murray, Sen. Maria Cantwell, and Rep. Susan DelBene, asking them to support Medicare for All and oppose Medicare Direct Contracting and privatization. Chapter leaders signed ten Washington State organizations onto the anti-Direct Contracting sign-on letter that was sent to HHS Sec. Becerra in early March. PNHP members also worked to pass a resolution in the Washington State Medical Association expressing support for “universal access to comprehensive, affordable, high-quality health care … including a publicly-funded national health care program.” The resolution passed with 93% of the vote. To get involved in Washington, contact Dr. McLanahan at mcltan@comcast.net.
In Wisconsin, the Linda and Gene Farley chapter has been active in PNHP’s campaign against Medicare Direct Contracting. In early February, the chapter hosted Dr. Ana Malinow to speak about DCEs with many allied organizations at the Wisconsin Health Matters coalition meeting. After the forum, several groups signed onto PNHP’s organizational letter to HHS Sec. Becerra. Chapter leaders also met with their Congressional representatives to educate them on Direct Contracting and Medicare privatization. To get involved in Wisconsin, contact wisconsin.pnhp@gmail.com.
“Physicians reflect on Martin Luther King Jr.’s legacy, efforts to combat health care inequities,” ABC News Los Angeles, 1/17/2022, featuring Drs. Paul Song and Susan Rogers
“Medicare is being privatized on Biden’s watch, insurance industry SALIVATES,” The Hill TV, 12/9/2022
On February 24, 2022, the Centers for Medicare and Medicaid Services (CMS) responded to criticism of its Direct Contracting pilot program by … changing the program’s name. CMS announced that Medicare Direct Contracting will come to a close at the end of 2022, and that it will be replaced by the virtually identical REACH model. This new incarnation retains the worst elements of the original program, including:
PNHP released a statement detailing our criticisms, and our president, Dr. Susan Rogers, issued a video response letting CMS know that we weren’t fooled by their transparent rebranding effort.
CMS claimed that REACH was developed after listening to feedback from concerned shareholders stakeholders, but all of the dangerous and insidious elements of the old program are still present in the new one. In fact, the REACH model also includes new giveaways to industry middlemen, and investors seem quite pleased with the announcement.
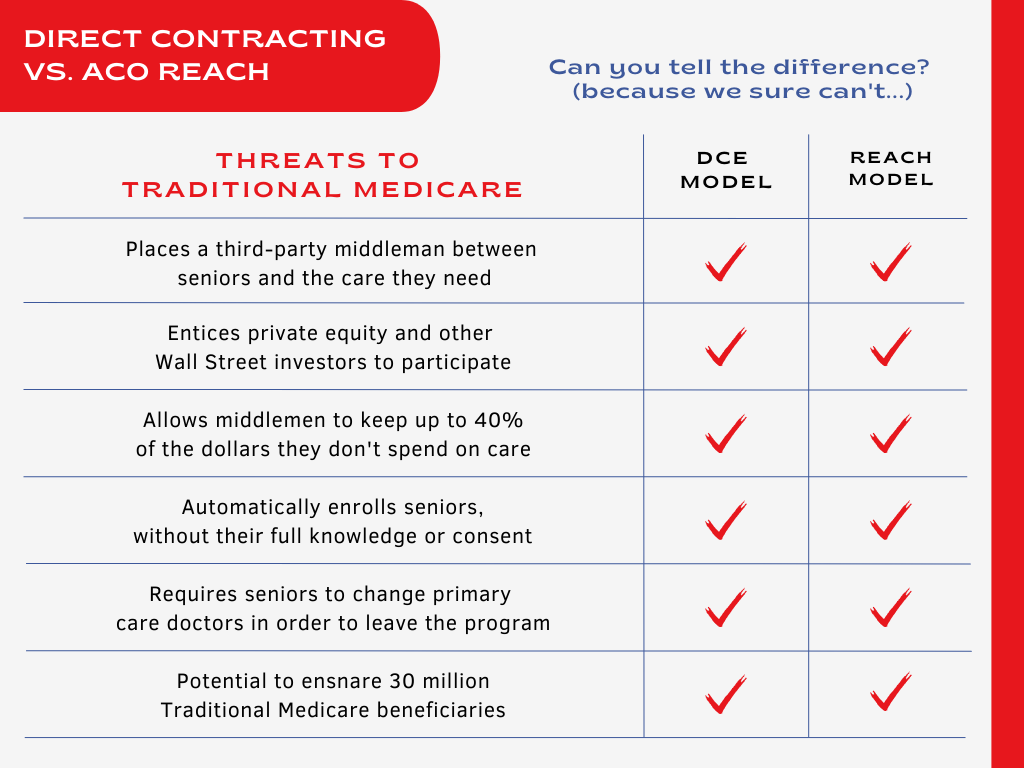
For more details on the newly announced REACH model, check out an 8:30 video from PNHP national board member Dr. Ed Weisbart, who explains the paper-thin nature of the changes offered by policymakers. Direct Contracting was broken beyond repair, and the “fixes” that CMS incorporated into REACH are wholly inadequate.
The only solution is to finally end this sneaky attempt to privatize Traditional Medicare.
Stay tuned as this story develops. In the meantime, be sure to sign and share our petition against Medicare privatization; call your member of Congress at (202) 224-3121; and educate your friends, family members, and colleagues about this ongoing threat to America’s seniors.
Physicians for a National Health Program (PNHP) advocates for a universal, comprehensive single-payer national health program (also called “Improved Medicare for All”). Since 1988, we’ve advocated for this reform in the U.S. health care system, growing into a nationally influential voice on health care reform with chapters and activists across the country, and a strong and growing student movement.
PNHP is a member-led organization that performs groundbreaking research on the health crisis and the need for fundamental reform, and uses a combination of popular education, policy expertise, and organizing to move the U.S. closer to single-payer health care.
PNHP, a national nonprofit organization based in Chicago, seeks a Policy and Communications Specialist to support our work to grow the single-payer health reform movement and put it at the top of the national and state agendas. The position is based in Chicago.
Position summary
The Policy and Communications Specialist works with the Executive Director and our Digital Communications Specialist to plan, initiate, execute, and manage public relations and member engagement activities for PNHP with the goals of (1) winning advocacy campaigns that bring us closer to public health care for everybody; (2) reaching influential figures in the medical profession, media, and government; (3) recruiting more members; and (4) increasing fundraising.
PNHP is a national authority on single-payer health reform and its communications staff must be able to understand and synthesize complex health policy issues, as well as how to communicate those issues to the media, advocates, and others in a wide range of formats including policy reports, media advisories, fact sheets, slide shows, email blasts, and calls to action.
On PNHP’s two-person communications team, this position leads on developing and producing policy materials, writing internal and external communications, engaging with reporters for traditional media outlets, and developing the media and spokesperson skills of PNHP’s leaders and chapters. The position will work closely with PNHP’s other Communications Specialist, who leads efforts on digital media.
Job duties
Developing policy tools and infrastructure (50% of time)
Communicating with PNHP members and supporters (25% of time)
Media Outreach and Response (10% of time)
Membership Development (10% of time)
Other duties as assigned (5% of time)
Qualifications and attributes
Salary and benefits
Salary: $55,000
Excellent benefits package including fully paid family comprehensive medical (platinum level HMO or PPO), dental and vision insurance; PTO.
Submission requirements
Email your resume along with a cover letter expressing your specific interest in PNHP and a writing sample to jobs@pnhp.org with the subject line “Communications Specialist Application First Name Last Name.” No calls please.
The anticipated start date for this position is May 15, 2024.
Persons of color, women, LGBTQ-identifying persons, and others from underrepresented groups are strongly encouraged to apply.
As the Biden administration continues to inexplicably roll out the Trump-era Medicare Direct Contracting program, PNHP members have been writing op-eds and letters to the editor demanding an end to this stealth privatization attempt. Members have also been appearing on television programs, radio shows, online video segments, and podcasts.
Interested in developing your own letter or op-ed? See our detailed suggestions on writing and submitting, or contact PNHP communications specialist Clare Fauke at clare@pnhp.org.
“Medicare is being privatized on Biden’s watch, insurance industry SALIVATES,” by Ryan Grim, Hill.TV, Rising, December 9, 2021
“Secret Trump Program To Privatize Medicare Needs to End,” interview with Dr. Susan Rogers, The Thom Hartmann Program, May 2, 2022
“Seniors’ Medicare Benefits are Being Privatized Without Consent,” The Lever, April 11, 2022
“The Stealth Plan to Corporatize Medicare,” interview with Dr. Ed Weisbart, The Zero Hour with RJ Eskow, March 11, 2022
“They’re Trying to Privatize Medicare!” interview with Diane Archer, The Zero Hour with RJ Eskow, January 19, 2022
“Direct Contracting Entities, Threat to our Healthcare,” interview with Drs. Claire Cohen and Marilyn Vaché, Thinking Green, January 11, 2022
“The Plot to Destroy Medicare,” interview with Dr. Ana Malinow, Thom Hartmann Program, December 13, 2021
“Is There a New Plot to Privatize Medicare Afoot?” interview with Dr. Ana Malinow, Thom Hartmann Program, September 20, 2021
“A Banker in the Exam Room? The Growing Resistance to Medicare Privatization,” interview with Dr. Ed Weisbart, Code Wack Podcast, March 28, 2022
“The end of traditional Medicare as we know it?” interview with Dr. Ed Weisbart, Retirement Revised Podcast, February 28, 2022
“Wall Street’s Latest Attack on Traditional Medicare,” interview with Dr. Ed Weisbart, Code Wack Podcast, October 18, 2021, (part one of this two-part podcast, which covers Medicare Advantage, available HERE)
“The Plot to Take Away Your Medicare, Unveiled!” interview with Dr. Ana Malinow, KSQD Community Radio, Be Bold America!, May 22, 2022
“In the Know: Medicare Direct Contracting,” interview with Dr. Ed Weisbart, KTRS The Big 550 AM, February 28, 2022
“The Corporate Assault on Medicare,” interview with Kip Sullivan, Ralph Nader Radio Hour, January 15, 2022
“The multiple dangers of Direct Contracting Entities,” interview with Kay Tillow, KPFA Radio, Flashpoints, December 2, 2021
“Medicare Direct Contracting call-in,” interview with Dr. Ana Malinow, Redwood Community Radio, KMUD, October 1, 2021
“The biggest threat to Medicare you’ve never even heard of,” by Dr. Susan Rogers and Rep. Pramila Jayapal, The Hill, December 9, 2021
PNHP president Dr. Susan Rogers co-authored a piece with H.R. 1976 (Medicare for All) lead sponsor Rep. Pramila Jayapal describing the dangers of Medicare Direct Contracting, and the urgent need to end the program.
“As a physician and a member of Congress, we’ve never heard a senior ask for their health care to be more complicated, or to have their choice of Traditional Medicare taken away,” they wrote. “But that’s exactly what Direct Contracting would do. Traditional Medicare has proven its value for more than half a century. Instead of selling it off to the highest bidder, let’s strengthen and improve its benefits while working to expand it to cover every American.”
One of the most outrageous features of Medicare Direct Contracting (and there are many) is the fact that it would make radical changes to Traditional Medicare, without so much as a vote in Congress. That’s because DCEs originated in the typically under-the-radar, and unaccountable, Center for Medicare and Medicaid Innovation.
Thankfully, elected officials are putting pressure on Health and Human Services Sec. Xavier Becerra to end Direct Contracting, which he has the power to do. This Trump-era program poses a grave threat to seniors and to Medicare as a whole, and it must be stopped immediately.
H.R. 1976 (Medicare for All) lead sponsor Rep. Pramila Jayapal and her staff have worked closely with PNHP and other health justice organizations to push back against DCEs, both publicly and within the halls of Congress.
PNHP thanks the following 54 members of the U.S. House of Representatives for signing on to Rep. Jayapal’s letter demanding an end to Direct Contracting:
Rep. Katie Porter has led on the issue of Direct Contracting from the beginning, having sent an earlier letter to Sec. Becerra (co-signed by Reps. Mark Pocan, Bill Pascrell, Jr., and Lloyd Doggett) expressing concern about the program and calling for an immediate freeze.
She joined a webinar that PNHP co-hosted with National Single Payer on Sept. 23, 2021, and warned viewers about the dangers of letting corporate interests infiltrate a cherished and effective public health program.
“This isn’t hypothetical,” she said. “We know exactly what happens when insurers dip their hands into Medicare, because we’ve already seen that play out in Medicare Advantage.”
Speaking of Medicare Advantage, former Centers for Medicare and Medicaid Services (CMS) Administrator Dr. Don Berwick and former Center for Medicare and Medicaid Innovation Director Dr. Rick Gilfillan authored a crucial two-part Health Affairs blog titled, “Medicare Advantage, Direct Contracting, and the Medicare ‘Money Machine.’”
“Given an Orwellian title, Direct Contracting, launched by CMMI, was anything but direct,” they wrote. “‘Indirect Contracting’ would have been a far more accurate name, since the cornerstone of the program was CMS’s opening the door to non-provider-controlled ‘Direct Contracting Entities (DCEs)’ to become the fiscal intermediaries between patients and providers.”
During the final days of the Trump Administration, officials at the Center for Medicare and Medicaid Innovation (CMMI) launched a dangerous and insidious pilot program known as Medicare Direct Contracting. (This program was recently rebranded as REACH by the Biden Administration.)
Left unchecked, Direct Contracting and REACH would allow profit-hungry middlemen to “manage” the care of up to 30 million Traditional Medicare beneficiaries. These middlemen, some of whom have received a huge influx of money from private equity, have every incentive to frustrate patients and deny medically necessary care.
Unless we stop REACH, this program could spell the end of Medicare as a public, nonprofit, social insurance program.

On February 2, 2022, PNHP president Dr. Susan Rogers testified before the U.S. Senate Committee on Finance, Subcommittee on Fiscal Responsibility and Economic Growth, regarding the dangers of Direct Contracting.
As Dr. Rogers stated in her testimony, “Medicare was designed as a lifeline for America’s seniors and those with disabilities, NOT a playground for Wall Street investors.”
On November 30, 2021, PNHP president Dr. Susan Rogers led a delegation to the doorsteps of the Dept. of Health and Human Services in Washington, D.C. to deliver 13,000+ petitions demanding an end to Medicare Direct Contracting.
If you’d like to JOIN our fight against Medicare Direct Contracting, please call your member of Congress at (202) 224-3121 and ask them to demand HHS end this dangerous and insidious program; hold hearings on DCEs; and establish Congressional oversight of the Center for Medicare and Medicaid Innovation (CMMI). You can also:
For the most current information on Direct Contracting and REACH, visit ProtectMedicare.net. To access archival information from the late 2021 and early 2022 phase of this fight, see the following pages:
Our live sessions took place October 22-23, 2021. For those who did not register in advance, we are offering $10 tickets that include a password for post-meeting access to archived content; click HERE to purchase.
Click the links below to jump to different sections of the newsletter. To view a PDF version of the shorter print edition of the newsletter, click HERE.
If you wish to support PNHP’s outreach and education efforts with a financial contribution, click HERE.
If you have feedback about the newsletter, email info@pnhp.org.
Moving organized medicine to Medicare for All should be a top priority

Polling shows that a majority of physicians support single-payer reform, with even stronger support among medical students and early-career physicians. Unfortunately, the vast majority of professional associations that claim to represent physicians do not support Medicare for All, and in some cases actively oppose it.
The lack of support by organized medicine is a huge problem for the Medicare-for-All movement. Medical societies have enormous influence over health care policy at the federal and state level. With the insurance, pharma, and investor-owned hospital industries spending millions each year lobbying against single-payer reform, the Medicare-for-All movement needs the full force of organized medicine fighting back.
As the only physician organization dedicated to single-payer reform, PNHP has a unique responsibility to move the medical profession to support Medicare for All. We’re meeting that challenge by launching the Medical Society Resolutions campaign.
“We can no longer ignore the elephant in the room — the powerful medical societies standing in the way of Medicare for All,” said PNHP president Dr. Susan Rogers. “As physicians, we are the only ones who can organize our colleagues for change.”
What is the Medical Society Resolutions (MSR) Campaign?
Virtually every physician is a member of a local, state, or specialty medical society, and is therefore in a position to propose resolutions that determine what policies their society will support (or oppose). The goal of the MSR campaign is to organize colleagues in the medical profession by passing Medicare-for-All resolutions in every medical society in the U.S.
The MSR campaign follows two years of unprecedented movement towards Medicare for All within organized medicine. In 2019, the American Medical Association only narrowly rejected a student-led pro-single payer resolution, opening the door for future support. Under pressure from activists, the AMA then resigned from the anti-single payer Partnership for America’s Health Care Future. In January 2020, the 159,000-member American College of Physicians — the largest medical specialty society and second-largest physician group after the AMA — announced its endorsement of Medicare for All, along with a “universal public choice” reform model. The 3,300-member Society of General Internal Medicine followed suit by formally endorsing the ACP’s position.
At the state level, the Vermont Medical Society overwhelmingly endorsed a single-payer resolution in November 2020, becoming the second state society to do so after Hawaii.
All the major associations representing medical students and new physicians have endorsed Medicare for All, including the American Medical Students Association; the AMA Medical Student Section; the Student National Medical Association (representing medical students of color); and the Committee of Interns and Residents.
How to participate in the MSR campaign
Every physician member of PNHP can participate in the MSR campaign by visiting medicalsocietyresolutions.org, where they can see if other PNHP members are actively organizing single-payer resolutions in their state, national, or specialty societies; download and edit a sample resolution; and watch recorded workshops on the nuts and bolts of passing resolutions. Interested members can contact organizer@pnhp.org to get started and connect with other PNHP members in their societies.
The U.S. is facing a crisis in maternal mortality. The rate of pregnancy-related deaths in the U.S. has nearly tripled in the past 30 years. Besides the U.S., the only other countries with rising maternal mortality rates are Afghanistan and Sudan. Our maternal mortality rate is more than double that of other high-income nations, and our infant mortality rate is 71% higher. Indigenous mothers are twice as likely — and Black mothers 2.5 times more likely — to die from pregnancy complications compared to white mothers.
The most frustrating aspect of this crisis is that these deaths are mostly preventable. Not with high-tech interventions, but with routine primary care. A majority of pregnancy-related complications and deaths are caused by health conditions that can be identified, managed, or even eliminated with regular preventive care, such as diabetes, heart disease, and hypertension. But addressing these conditions before pregnancy requires a lifetime of regular care, which is out of reach for many Americans — especially people of color — because of costs and insurance barriers.
We won’t solve this crisis with band-aid approaches. Even Medicaid — which now finances nearly half the births in the U.S. — only starts after a person discovers they are pregnant, and in many states ends soon after birth, limiting access to critically important prenatal and postpartum care.
By providing lifelong coverage for all medically necessary health services, including primary care; mental health; contraception and abortion; and pregnancy, childbirth, and postpartum care — Medicare for All is the only plan that would empower patients to prevent, identify, and treat the chronic health conditions that increase risk of pregnancy-related complications and death.
Visit pnhp.org/maternalmortality to explore the full toolkit on America’s maternal mortality crisis, which includes talking points and handouts, a slide set and webinar, videos and podcasts, and a new interactive quiz to test your knowledge of this crisis. Please use these materials in your own advocacy and share with your colleagues and communities. To learn more about PNHP’s Kitchen Table Campaign, contact clare@pnhp.org.
 Sanjeev Sriram, M.D., M.P.H. has been a PNHP member since 2018. Dr. Sriram completed his medical degree and residency at UCLA, his masters in public health at Harvard, and now practices pediatrics in Maryland. He founded the “All Means All” campaign to center racial equity in single payer, and promotes Medicare for All and health equity in national publications and as “Dr. America” for act.TV. As a board member, Dr. Sriram will urge PNHP to collaborate with racial justice organizations on strategies to broaden single-payer activism, and expand mentorship and professional development opportunities for our student members.
Sanjeev Sriram, M.D., M.P.H. has been a PNHP member since 2018. Dr. Sriram completed his medical degree and residency at UCLA, his masters in public health at Harvard, and now practices pediatrics in Maryland. He founded the “All Means All” campaign to center racial equity in single payer, and promotes Medicare for All and health equity in national publications and as “Dr. America” for act.TV. As a board member, Dr. Sriram will urge PNHP to collaborate with racial justice organizations on strategies to broaden single-payer activism, and expand mentorship and professional development opportunities for our student members.
 Philip Verhoef, M.D., Ph.D. has been a PNHP member since 2006. Dr. Verhoef received his medical degree and a Ph.D. in pharmacology from Case Western Reserve University, followed by med-peds residency at UCLA. He joined the faculty at the University of Chicago after a fellowship in adult and pediatric critical care, while serving as the president of the Illinois Single Payer Coalition and co-president of PNHP-Illinois. In 2019, Dr. Verhoef moved to Kaiser Permanente in Hawaii, where he serves as an adult/pediatric intensivist and hospitalist and as the associate program director for the internal medicine residency program. As a continuing board member, he plans to develop strategies for physician engagement, including messaging, programming, development, and recruitment, with a specific focus on diverse and underserved communities.
Philip Verhoef, M.D., Ph.D. has been a PNHP member since 2006. Dr. Verhoef received his medical degree and a Ph.D. in pharmacology from Case Western Reserve University, followed by med-peds residency at UCLA. He joined the faculty at the University of Chicago after a fellowship in adult and pediatric critical care, while serving as the president of the Illinois Single Payer Coalition and co-president of PNHP-Illinois. In 2019, Dr. Verhoef moved to Kaiser Permanente in Hawaii, where he serves as an adult/pediatric intensivist and hospitalist and as the associate program director for the internal medicine residency program. As a continuing board member, he plans to develop strategies for physician engagement, including messaging, programming, development, and recruitment, with a specific focus on diverse and underserved communities.
Continuing at-large member: Stephen Chao, M.D. (Texas)
 Scott Goldberg, M.D. has been a PNHP member since 2009. Dr. Goldberg earned his medical degree at University of Chicago Pritzker School of Medicine (where he launched a SNaHP chapter in 2012) and completed residency in 2019 in internal medicine-primary care at UCSF. He serves as an attending physician at Montefiore Medical Center in the Bronx where he supervises residents in the Primary Care/Social Internal Medicine program, and an assistant professor at the Albert Einstein College of Medicine. As a board member, Dr. Goldberg plans to expand PNHP’s fundraising and membership efforts, with the goal of building the power necessary to shape the culture of organized medicine around single payer.
Scott Goldberg, M.D. has been a PNHP member since 2009. Dr. Goldberg earned his medical degree at University of Chicago Pritzker School of Medicine (where he launched a SNaHP chapter in 2012) and completed residency in 2019 in internal medicine-primary care at UCSF. He serves as an attending physician at Montefiore Medical Center in the Bronx where he supervises residents in the Primary Care/Social Internal Medicine program, and an assistant professor at the Albert Einstein College of Medicine. As a board member, Dr. Goldberg plans to expand PNHP’s fundraising and membership efforts, with the goal of building the power necessary to shape the culture of organized medicine around single payer.
Continuing North East board members: Mary O’Brien, M.D. (New York) and Janine Petito, M.D. (Massachusetts)
 Ed Weisbart, M.D. has been a PNHP member since 1996. He completed his medical degree at the University of Illinois and family medicine residency at Michigan State University. Dr. Weisbart practiced family medicine for 20 years, served as CMO of Express Scripts from 2003-2010, and retired clinically in 2021. He serves as the chair of the Missouri chapter of PNHP, where he has delivered more than 600 public presentations and published dozens of pieces on single payer. As a board member, Dr. Weisbart will help PNHP increase its influence within the health care policy debate, and strengthen its relationships with adjacent advocacy organizations.
Ed Weisbart, M.D. has been a PNHP member since 1996. He completed his medical degree at the University of Illinois and family medicine residency at Michigan State University. Dr. Weisbart practiced family medicine for 20 years, served as CMO of Express Scripts from 2003-2010, and retired clinically in 2021. He serves as the chair of the Missouri chapter of PNHP, where he has delivered more than 600 public presentations and published dozens of pieces on single payer. As a board member, Dr. Weisbart will help PNHP increase its influence within the health care policy debate, and strengthen its relationships with adjacent advocacy organizations.
Continuing South Region board member: Jessica Schorr Saxe, M.D. (North Carolina)
 Judith Albert, M.D.> has been a PNHP member since 2017. She received her medical degree from the University of Cincinnati, completed residency in obstetrics and gynecology at the University of Pittsburgh and fellowship in reproductive endocrinology at the University of Pennsylvania. Dr. Albert recently retired from having practiced in academic and private practice settings for over 30 years in Pittsburgh. She co-founded a PNHP chapter in western Pennsylvania where she has been involved in anti-racist organizing for several years. As a board member, Dr. Albert hopes to grow the single-payer movement by strengthening PNHP chapters in the region, and building coalitions with anti-racist, fair housing and anti-poverty organizations, as well as labor unions.
Judith Albert, M.D.> has been a PNHP member since 2017. She received her medical degree from the University of Cincinnati, completed residency in obstetrics and gynecology at the University of Pittsburgh and fellowship in reproductive endocrinology at the University of Pennsylvania. Dr. Albert recently retired from having practiced in academic and private practice settings for over 30 years in Pittsburgh. She co-founded a PNHP chapter in western Pennsylvania where she has been involved in anti-racist organizing for several years. As a board member, Dr. Albert hopes to grow the single-payer movement by strengthening PNHP chapters in the region, and building coalitions with anti-racist, fair housing and anti-poverty organizations, as well as labor unions.
Continuing North Central Region board member: Joshua J. Faucher, M.D., J.D. (Illinois)
 Kathleen Healey, M.D. has been a PNHP member since 2018. Dr. Healey completed her medical degree at University of Colorado School of Medicine and her residency at the Naval Medical Center in Oakland. She is an otolaryngologist whose career ranged from military service as a flight surgeon, to solo and group practices. Now retired, Dr. Healey serves as co-chair of PNHP-Napa County and PNHP-California. As a board member, Dr. Healey will work to bring more physicians into our movement, and increase PNHP’s involvement in organized medicine at all levels.
Kathleen Healey, M.D. has been a PNHP member since 2018. Dr. Healey completed her medical degree at University of Colorado School of Medicine and her residency at the Naval Medical Center in Oakland. She is an otolaryngologist whose career ranged from military service as a flight surgeon, to solo and group practices. Now retired, Dr. Healey serves as co-chair of PNHP-Napa County and PNHP-California. As a board member, Dr. Healey will work to bring more physicians into our movement, and increase PNHP’s involvement in organized medicine at all levels.
 Stephen Kemble, M.D. has been a PNHP member since 1989. Dr. Kemble attended medical school at University of Hawaii and Harvard, and completed residencies in both internal medicine (Queen’s Medical Center) and psychiatry (Cambridge Health Alliance). He is now semi-retired, working part-time in a primary care clinic. Dr. Kemble is past president of both the Hawaii Psychiatric Medical Association and the Hawaii Medical Association, and also served on the Hawaii Health Authority board. As a board member, Dr. Kemble will continue to chair the newly formed PNHP Policy Committee, and help PNHP fight the power of the insurance industry with ongoing public education and community organizing.
Stephen Kemble, M.D. has been a PNHP member since 1989. Dr. Kemble attended medical school at University of Hawaii and Harvard, and completed residencies in both internal medicine (Queen’s Medical Center) and psychiatry (Cambridge Health Alliance). He is now semi-retired, working part-time in a primary care clinic. Dr. Kemble is past president of both the Hawaii Psychiatric Medical Association and the Hawaii Medical Association, and also served on the Hawaii Health Authority board. As a board member, Dr. Kemble will continue to chair the newly formed PNHP Policy Committee, and help PNHP fight the power of the insurance industry with ongoing public education and community organizing.
Continuing West Region board member: Eve Shapiro, M.D., M.P.H. (Arizona)
Both the insured and uninsured struggle with medical costs. Nearly one in four (38%) adults had medical bill or debt problems in the last year, including 46% of those on individual/marketplace plans, 34% of those on employer plans, 55% of Black people, and 47% of low-income people; a third of those with debt said they were paying off $4,000 or more. Among those with medical debt problems, 35% used up all or most of their savings, 35% took on credit card debt, 27% had been unable to pay for basic necessities like food or rent, 23% delayed education or career plans, and 43% received a lowered credit score. Although uninsured people reported medical bill problems at the highest rates, 64% of those with a medical bill or debt problem said they had been insured at the time. Collins et al., “As the pandemic eases, what is the state of health care coverage and affordability in the U.S.? Findings from the Commonwealth Fund health care coverage and Covid-19 survey, March–June 2021,” Commonwealth Fund, 7/16/2021
Even high-income Americans have trouble paying for care. In the past year, nearly one in four Americans (38%) said they had trouble accessing health care because of cost, including 27% of high-income earners; 36% skipped health or dental care because of cost, including 21% of high-income earners; 34% said their insurance denied payment for medical care; 22% had serious problems paying or were unable to pay medical bills; and 44% had out-of-pocket medical expenses exceeding $1,000. Schneider et al., “Mirror, Mirror 2021 — Reflecting poorly: Health care in the U.S. compared to other high-income countries,” Commonwealth Fund, 8/4/2021
Americans’ medical debt reaches record levels. An estimated 17.8% of individuals in the U.S. had medical debt in collections in June 2020, for care provided prior to the pandemic. Collection agencies held $140 billion in unpaid medical bills, up from an estimated $81 billion in 2016. Between 2009 and 2020, unpaid medical bills became the largest source of debt that Americans owed to collection agencies. Residents of states that did not expand Medicaid owed an average of $375 more compared to those in expansion states, roughly a 30% increase from the year before Medicaid expansion. People living in the lowest-income ZIP codes owed an average of $677, compared to $126 in the highest-income ZIP codes. Kluender et al., “Medical debt in the U.S., 2009-2020,” JAMA, 7/20/2021
Providing medications for free leads to greater adherence and cost savings. In a study of patients in Ontario who reported cost-related non-adherence to medications, providing those medications for free increased patient adherence by 35% and reduced total health spending, including hospitalization, by an average of $1,222 per patient per year. Persaud et al., “Adherence at two years with distribution of essential medicines at no charge: The CLEAN Meds randomized clinical trial,” PLOS Medicine, 5/21/2021
U.S. life expectancy drops most for people of color. Life expectancy in the U.S. decreased by nearly two years between 2018 and 2020, down to 76.9 at the end of 2020 from 78.7 in 2018. However, the declines were more pronounced among Black people, whose life expectancy decreased by 3.3 years, and Latinx/Hispanic people, whose life expectancy decreased by 3.9 years. By comparison, among a group of 16 peer countries, the average decline in life expectancy was 0.22 years (about two-and-a-half months). Woolf et al., “Effect of the Covid-19 pandemic in 2020 on life expectancy across populations in the USA and other high-income countries: Simulations of provisional mortality data,” BMJ, 5/24/2021
Life expectancy gap widens between urban and rural communities. In 2019, the mortality rate in urban areas of the U.S. was nearly 665 deaths per 100,000 people, but in rural areas was 834 deaths per 100,000 people. Over the past 20 years, the life expectancy gap between rural and urban areas grew by 172%. Cross et al., “Rural-urban disparity in mortality in the U.S. from 1999 to 2019,” JAMA Network, 6/8/2021
Latinx/Hispanic children in the U.S. are twice as likely to be uninsured. The uninsured rate for Latinx/Hispanic children in the U.S. reached 9.3% in 2019, compared to an uninsured rate of 4.4% for non-Latinx/Hispanic youth. There is considerable variation in the uninsured rate based on state, ranging from 1.8% uninsured in Massachusetts to 19.2% in Mississippi. The uninsured rate for Latinx/Hispanic children in states that had not expanded Medicaid by 2019 was more than 2.5 times higher than expansion states (14.9% vs. 5.8%). Whitener and Corcoran, “Getting back on track: A detailed look at health coverage trends for Latino children,” Georgetown University Center for Children and Families, 6/8/2021
U.S. health spending goes disproportionately to white patients. The U.S. spends about 15% more on health services for white people than for people of color. Per-person spending for white people averaged $8,141, compared to $7,361 for Black people, $6,025 for Latinx/Hispanic people, and $4,692 for Asian, Native Hawaiian, and Pacific Islander people. Spending also differed by types of care. For example, compared to the national average, Black people accounted for 26% less spending on outpatient services, but 12% more on emergency or inpatient care, suggesting they are treated for illnesses at more advanced stages. Dieleman et al., “U.S. health care spending by race and ethnicity, 2002-2016,” JAMA Network, 8/17/2021
Low-cost care improves colon cancer survival rates, especially for Black patients. Between 1987 and 2013, colon cancer patients in the U.S. Military Health System (MHS) — where care is provided with few or no financial barriers — had an 18% lower risk of death, and were 10% less likely to be diagnosed in a later phase of the disease, compared to similar patients in the general population. The better survival rates were also more evident among Black patients in the MHS, who were 26% less likely to die of colon cancer than those in the general population. Lin et al., “Comparison of survival among colon cancer patients in the U.S. Military Health System and patients in the Surveillance, Epidemiology, and End Results (SEER) Program,” Cancer Epidemiology, Biomarkers & Prevention, 6/23/2021
Medicare coverage reduces racial disparities in coverage and care. Eligibility for Medicare coverage was associated with reductions in racial and ethnic disparities in insurance coverage, access to care, and self-reported health, benefiting Black and Latinx/Hispanic people the most. Medicare eligibility shrank disparities in insurance coverage by 53% between Black and white people, and 51% between Latinx/Hispanic and white people. Insurance coverage for Latinx/Hispanic people rose from 77.4% prior to the age of 65 to 91.3% after 65; for Black people, it rose from 86.3% to 95.8%. The proportion of Black and Latinx people who self-reported their health as poor also dropped significantly after they became eligible for Medicare. Wallace et al., “Changes in racial and ethnic disparities in access to care and health among U.S. adults at age 65 years,” JAMA Internal Medicine, 7/26/2021
Workplace “wellness” programs are no substitute for actual health care. A controlled study of workplace wellness programs — which included modules on nutrition, physical activity, and stress reduction — found that employees at the wellness program worksites had better self-reported health behaviors (such as attempting to manage their weight), but found no significant differences in self-reported health, clinical markers of health, health care spending or use, absenteeism, tenure, or job performance. A three-year follow up did not yield detectable improvements in clinical, economic, or employment outcomes. Song and Baicker, “Health and economic outcomes up to three years after a workplace wellness program: A randomized controlled trial,” Health Affairs, June 2021
High-deductible health plans (HDHP) now the norm. The majority (51%) of private-sector employees are now enrolled in HDHPs, defined as having a deductible of $1,350 for an individual and $2,700 for a family in 2018. HDHP enrollment has grown by 43% over the past five years. “State health compare,” State Health Access Data Assistance Center, Health Policy and Management Division of the School of Public Health at the University of Minnesota, accessed August 2021
High cost-sharing has potentially deadly consequences for lower-income patients. Among patients who had been forced to switch from a low-deductible to a high-deductible health plan (HDHP), researchers found that patients from low-income neighborhoods had fewer emergency department visits or hospitalizations for nonspecific chest pain, but had more hospitalizations for myocardial infarction (heart attack) after ED diagnosis of nonspecific chest pain. Researchers conclude that HDHPs’ higher out-of-pocket costs lead to potentially negative health implications for lower-income populations. Chou et al., “Impact of high-deductible health plans on emergency department patients with nonspecific chest pain and their subsequent care,” Circulation, June 2021
Patients again saddled with Covid care cost-sharing burden. Despite record profits and a recent surge in Covid cases, insurers are dropping their Covid-19 cost-sharing waivers. Across the two largest health plans in each state, 72% are no longer waiving out-of-pocket costs for Covid-19 treatment, with another 10% phasing out their waiver policies by the end of October. Almost half these plans ended cost-sharing waivers in April 2021, citing vaccine availability. Ortaliza et al., “Most private insurers are no longer waiving cost-sharing for Covid-19 treatment,” Kaiser Family Foundation, 8/19/2021
Those with commercial insurance face worse access, higher costs than those in public plans. Compared to people on Medicare, those with employer-sponsored or “marketplace” plans were less satisfied with their care, less likely to have a personal physician, and more likely to report instability in insurance coverage and difficulty receiving medical care or prescriptions due to cost. Reports of medical debt were more common among people who had employer-sponsored coverage (23.4%) and those with individual commercial plans (22.3%) than individuals covered by Medicare (15.6%) or Medicaid (18.3%). Wray et al., “Access to care, cost of care, and satisfaction with care among adults with private and public health insurance in the U.S.,” JAMA, 6/1/2021
Commercial plans save money by denying patient claims. Insurers offering individual ACA “marketplace” plans denied about 17% of in-network claims (40.4 million) in 2019. Patients almost never appeal claim denials: 0.2% of patients appealed their denials, vs. 99.8% that did not appeal. When patients did appeal, insurers upheld 60% of those denials. Even though patients have the right to request an external review after a claims appeal is denied by the insurer, fewer than one in 20,000 denied claims made it to external review. Pollitz and McDermott, “Claims denials and appeals in ACA Marketplace plans,” Kaiser Family Foundation, 1/20/2021
Insurers’ “utilization management” schemes cost the health care system $93 billion per year, with patients paying most of the cost. Insurers are restricting drug formularies, requiring more stringent prior authorizations, and raising patient cost-sharing requirements for prescriptions. These so-called “utilization management” schemes cost the U.S. health system approximately $93.3 billion each year for implementing, contesting, and navigation. Insurers spend approximately $6.0 billion administering utilization management, and drug companies spend approximately $24.8 billion subsidizing patient copays. However, the biggest costs are borne by patients and doctors: Physicians waste $26.7 billion on time spent navigating utilization management, and patients spend $35.8 billion in drug cost sharing, even after copay coupons from manufacturers and charities. The study did not measure the health effects of these schemes, but notes that approximately 20% of prescriptions in the U.S. are never filled. Howell et al., “Quantifying the economic burden of drug utilization management on payers, manufacturers, physicians, and patients,” Health Affairs, August 2021
Insurers gobble up provider practices and keep more of patients’ premiums. Some commercial insurers are expanding aggressively into care delivery, and get to keep more of the premiums they collect when they also own the providers. Federal law limits insurers’ profits to 15-20% of collected premiums, but puts no limits to how much profit a provider can keep. So if an insurer directs enrollees to insurer-owned providers, the company is able to keep more premium dollars. UnitedHealth, for example, owns commercial insurance plans but also operates Optum-branded surgery centers, physician practices, and specialty pharmacies. In 2021, UnitedHealth expects to earn $91 billion in “eliminations,” an accounting term for revenues that stay within the company, a fourfold increase from 10 years ago. Herman, “Profits swell when insurers are also your doctors,” Axios, 7/16/2021
Not all surprise medical bills come from the hospital. Among large employer health plans in 2018, about half (51%) of emergency and 39% of non-emergency ground ambulance rides included an out-of-network charge for ambulance services, sticking patients with surprise bills. Ambulances bring 3 million privately insured people to an emergency room each year. Amin et al., “Ground ambulance rides and potential for surprise billing,” Peterson-Kaiser Family Foundation Health System Tracker, 6/24/2021
Commercial insurers continue pandemic profit streak. At the mid-point of 2021 — as the U.S. entered another wave of Covid hospitalizations and death — commercial insurers posted massive profits. UnitedHealth led the way with second quarter net profits of $4.3 billion; CVS Health (Aetna), $2.8 billion; Anthem, $1.8 billion; Cigna, $1.5 billion; and Humana, $588 million. Herman, “The vaccine wave kept health care as profitable as ever,” Axios, 8/30/2021
Medicare Advantage drives up Medicare spending. Medicare spending for enrollees on privatized Medicare Advantage (MA) plans was $321 higher per person in 2019 than it would have been if enrollees had been covered by traditional Medicare, raising overall Medicare spending by $7 billion. Between 2021 and 2029, federal spending on payments to MA plans is projected to increase by $316 billion, from $348 billion to $664 billion. Biniek et al., “Higher and faster growing spending per Medicare Advantage enrollee adds to Medicare’s solvency and affordability challenges,” Kaiser Family Foundation, 8/17/2021
Medicare Advantage plans find ways to dump dying patients. Commercial Medicare Advantage (MA) plans are finding ways to avoid paying the high costs of end-of-life care. As a result, MA beneficiaries in the last year of life disenrolled to join traditional Medicare at more than twice the rate (5%) of all other MA beneficiaries (2%) in 2017. The U.S. Government Accountability Office found that beneficiaries in the last year of life disenroll because of limitations accessing specialized (and expensive) care under MA. Because Medicare pays MA a fixed fee per enrollee, MA enrollees who switched to traditional fee-for-service Medicare in their last year of life increased Medicare’s costs by $490 million in 2017. “Medicare Advantage: Beneficiary disenrollments to fee-for-service in last year of life increase Medicare spending,” U.S. Government Accountability Office, 6/28/2021
Medicare enrollees face cost problems, but fare better in traditional Medicare than Medicare Advantage. The rate of cost-related problems was lower among beneficiaries in traditional Medicare (TM) (15%) than among those enrolled in Medicare Advantage (MA) (19%) plans. Those with TM plus supplemental coverage (80% of those in TM) had the lowest cost-related problems (12%), but among the remaining 20% of TM enrollees without supplemental coverage, 30% reported cost-related problems. Among Black beneficiaries, those in TM had lower cost problems (24%) than those in MA (32%). Across all plans, the rate of cost-related problems was twice as high among Black beneficiaries compared to white beneficiaries (28% vs. 14%), three times higher among beneficiaries in fair or poor self-reported health than among those good health (34% vs. 11%), and 3.5 times higher among beneficiaries under age 65 with long-term disabilities than among those ages 65 and older (42% vs. 12%). Biniek et al., “Cost-related problems are less common among beneficiaries in traditional Medicare than in Medicare Advantage, mainly due to supplemental coverage,” Kaiser Family Foundation, 6/25/2021
Despite the pandemic, health industry CEOs have big paydays. The CEOs of 178 health care companies collectively made $3.2 billion in 2020, 31% more than 2019. The CEOs of the six biggest commercial health insurers (Anthem, Centene, Cigna, CVS Health, Humana, UnitedHealth Group) made a combined $236 million in 2020, a 45% increase over 2019. Herman, “Health care executive pay soars during pandemic,” Axios, 6/14/2021
High-revenue hospitals more likely to sue their patients. More than a quarter of the 100 U.S. hospitals with the highest revenues sued patients over unpaid medical bills between 2018 and mid-2020, filing nearly 39,000 court actions (which is likely an undercount since many court records are inaccessible), including lawsuits, wage garnishments, and personal property liens. McGhee and Chase, “How America’s top hospitals hound patients with predatory billing,” Axios, 6/14/2021
Investor-owned hospitals more likely to inflate prices. Most hospitals charge more for a procedure than what it costs them, but for-profit facilities take this markup to extremes. The top 100 revenue-generating hospitals charged patients seven times the cost of service, and for-profit hospitals averaged a nearly 12-fold markup. Nine of the 10 top-markup hospitals were investor owned. While these charges are almost never the actual price paid by insurers, they are used to charge uninsured patients. McGhee and Chase, “How private hospitals make their money: Massive markups,” Axios, 6/14/2021
Investors are cashing in on trauma centers. Investor-owned hospital firms like HCA are rapidly opening “trauma centers,” which treat injuries from events like car crashes, falls, or gunshot wounds. Trauma centers were once operated mainly by established teaching hospitals, but investor-owned HCA has opened trauma centers in 90 of its hospitals and now operates one out of every 20 trauma centers in the country. Once a hospital has a trauma designation, it can charge patients special “trauma team activation” fees of as much as $50,000 per patient for the same care provided in a regular emergency department. Hancock, “In alleged health care ‘money grab,’ nation’s largest hospital chain cashes in on trauma centers,” Kaiser Health News, 6/14/2021
Independent physician practices are now the minority. By the end of 2020, hospitals and corporations owned half of America’s physician practices; nearly 70% of U.S. physicians are now employed by hospitals or corporations like private equity firms and health insurers. In 2019 and 2020, 48,000 physicians quit private practice; of those, more physicians moved to corporate entities than to hospitals. Corporate entities now employ an estimated 20% of all physicians, a 31% increase in the percentage of corporate-employed physicians over two years. “COVID-19’s impact on acquisitions of physician practices and physician employment 2019-2020,” Avalere Health, June 2021
Drug prices are rising at twice the rate of inflation. While the 2020 rate of inflation was 1.3%, the price of a group of 260 widely used prescription drugs rose by nearly 3% overall since 2019. Over the past 15 years, the price of 65 regularly used brand name drugs rose by nearly 280%, while inflation only rose by 32%. Purvis and Schondelmeyer, “Rx price watch report: Trends in retail prices of brand name prescription drugs widely used by older Americans, 2006 to 2020,” AARP Public Policy Institute, June 2021
Medicare drug spending spikes due to prices, not volume. The amount Medicare spent on drugs dispensed at pharmacies increased 26% from 2013 through 2018. The Medicare Payment Advisory Commission attributed nearly all of the growth in spending to higher prices charged by pharmaceutical firms rather than an increase in the number of prescriptions filled by beneficiaries. “Report to the Congress: Medicare and the health care delivery system,” The Medicare Payment Advisory Commission, June 2021
Direct advertising leads to increases in Medicare spending on expensive drugs. Pharma manufacturers spend about $6 billion each year on consumer advertising. The highest ad spenders were AbbVie’s rheumatoid arthritis drug Humira at $1.4 billion; Pfizer’s neuropathic pain drug Lyrica at $913 million; and Eli Lilly’s Type 2 diabetes drug Trulicity at $655 million. Between 2016 and 2018, nearly 60% of Medicare Parts B and D beneficiary spending ($324 billion) went to drugs the industry advertised directly to consumers. Advertised drugs accounted for 8% of total Medicare Part D drugs used but 57% of drug spending. Among the top 10 drugs with the highest Medicare expenditures, four were also among the top 10 drugs in advertising spending in 2018. “Prescription drugs: Medicare spending on drugs with direct-to-consumer advertising,” Government Accountability Office Report to U.S. Senate Committee on the Judiciary, May 2021
Pharma payments to doctors are associated with increased prescribing of more expensive insulin. An analysis of Medicare claims found that more than 51,800 physicians received industry payments worth $22.3 million in 2016. The following year, those physicians wrote, on average, 135 prescriptions for more expensive long-acting insulin, compared with 77 prescriptions written by doctors who did not receive industry payments. The larger number of prescriptions resulted in an average Medicare Part D claim of $300, which was $71 more than claims generated by doctors who did not receive payments. Inoue et al., “Association between industry payments and prescriptions of long-acting insulin: An observational study with propensity score matching,” PLOS Medicine, 6/1/2021
Pharma spends more on dividends and stock buybacks than research and development. The 14 largest drugmakers spent $577 billion on stock buybacks and dividends from 2016 through 2020, which was $56 billion more than was spent on R&D during the same time. In fact, some of the spending categorized as “research and development” was spent “researching” ways to suppress competition — especially from generics — such as filing hundreds of new but very minor patents on older drugs. “Drug pricing investigation: Industry spending on buybacks, dividends, and executive compensation,” U.S. House of Representatives Committee on Oversight and Reform, July 2021
Some of the biggest patient advocacy groups take millions from drug companies, but hide those relationships. All but one of the 15 most prominent patient advocacy organizations — including the American Cancer Society, American Diabetes Association, American Heart Association, and American Lung Association — fail to fully disclose the amount of drug industry funding they receive, and 12 of the 15 leading groups also have representation from the pharmaceutical industry on their boards. One of the groups, the International Myeloma Foundation, received 57% of its funding ($11.5 million) from just two pharmaceutical companies. Researchers found that many of these same groups “appear unable or unwilling to take positions on consumer issues such as lowering prescription drug prices that might anger their drug corporation funders.” “The hidden hand: Big pharma’s influence on patient advocacy groups,” Patients for Affordable Drugs, 6/30/2021
Pharma keeps prices high by buying off lawmakers. Nearly every attempt to lower drug prices at the state level has failed. Not surprisingly, more than one-third of state legislators in the U.S. (at least 2,467) took pharmaceutical industry campaign contributions in the last two years. In Louisiana 84% of lawmakers took cash from pharma during the 2020 election cycle; in California it was 82%; in Illinois, 76%; and in Oregon, 66%. The industry spent slightly more on Democrats ($4.5 million) than on Republicans ($4.4 million). Facher, “Pharma funded more than 2,400 state lawmaker campaigns in 2020, new STAT analysis finds,” STAT, 6/9/2021
Pharma throws cash at Dems who fight Medicare drug negotiations. In early May, Rep. Scott Peters (D-Calif.) led a group of nine centrist Democrats attempting to block Rep. Nancy Pelosi’s bill allowing Medicare to negotiate drug prices. Over the next two days, Rep. Peters received $19,600 from the pharmaceutical industry, including $5,800 from Pfizer CEO Albert Bourla, $5,000 from Eli Lilly CEO David Ricks, $2,900 from Bristol Myers Squibb CEO Giovanni Caforio, $2,900 from Merck CEO Ken Frazier, and three $1,000 checks from three separate PhRMA lobbyists. In total, Rep. Peters took in $66,400 from the pharmaceutical industry between May 4 and June 30. Cohrs, “Pharma CEOs, lobbyists showered Democrat with cash after his attempt to torpedo Pelosi’s drug pricing bill,” STAT, 7/20/2021
“Racial Justice Report Card, 2020-2021,” White Coats for Black Lives, September 1, 2021. The goal of the Racial Justice Report Card is to document the racism that continues to permeate our nation’s leading medical institutions, highlight best practices, and encourage academic medical centers to direct their considerable power and resources toward addressing the needs of patients and colleagues of color. The Report Card consists of metrics that evaluate medical institutions’ curriculum and climate, student and faculty diversity, policing, racial integration of clinical care sites, treatment of workers, and research protocols. All of the schools received mostly failing grades.
“Medical debt in the U.S., 2009-2020,” Raymond Kluender, Ph.D.; Neale Mahoney, Ph.D.; Francis Wong, Ph.D.; Wesley Yin, Ph.D.; JAMA Network, July 20, 2021. Between 2009 and 2020, unpaid medical bills became the largest source of debt that Americans owe collections agencies; by 2020, these agencies held $140 billion in unpaid medical bills. That amount only measures debts that have been sold to collectors and does not count all medical bills owed to health care providers. An estimated 17.8% of individuals in the U.S. had medical debt in collections in June 2020, reflecting care provided prior to the Covid-19 pandemic. Medical debt was highest among individuals who lived in the South and in ZIP codes in the lowest income deciles, and was most concentrated in lower-income communities in states that did not expand Medicaid.
“Medical uninsurance and underinsurance among U.S. Children: Findings from the National Survey of Children’s Health, 2016-2019,” Adam Gaffney, M.D., M.P.H.; Samuel Dickman, M.D.; Christopher Cai, M.D.; Danny McCormick, M.D., M.P.H.; David U. Himmelstein, M.D.; Steffie Woolhandler, M.D., M.P.H.; JAMA Pediatrics, August 23, 2021. Researchers found that between 2016-19, the number of uninsured children rose from 5.9 million to 6.3 million, and the number of children with inadequate coverage increased from 16.2 million to 18.1 million. The proportion of children with inadequate insurance (either uninsured or underinsured) was lower in Medicaid expansion states (30.9%) than in non-expansion states (35.3%). Underinsurance was more common among privately-insured (34.8%) than publicly-insured (17.5%) children, likely reflecting the high copayments and deductibles in many private plans. The researchers also found that nearly one in three children with serious chronic illnesses or impairments were inadequately insured.
“Association between high-deductible health plans and cost-related non-adherence to medications among Americans with diabetes: An observational study,” Charlotte Rastas, M.D., M.Sc.; Drew Bunker, M.D.; Vikas Gampa, M.D.; John Gaudet, M.D.; Shirin Karimi, M.D.; Ariel Majidi, M.P.; Gaurab Basu, M.D., M.P.H.; Adam Gaffney, M.D., M.P.H.; and Danny McCormick, M.D., M.P.H.; Journal of General Internal Medicine, July 29, 2021. For Americans with diabetes, being enrolled in a high-deductible health plan (HDHP) substantially increases the risk of not taking prescribed medications due to cost. The study found that among all patients with diabetes, 20% of those enrolled in a HDHP reported forgoing medications due to cost, compared with 16% of those in a traditional commercial plan — a 28% higher rate of missing medication for those with a high deductible. Among patients specifically taking insulin for diabetes, 25% of HDHP enrollees were unable to afford their medication, compared with 19% of those in a traditional plan — a 31% higher rate of missing medications. Researchers also found that among the diabetic patients they studied, those who could not take their medication as prescribed because they could not afford it were more likely to have one or more emergency department visits, and potentially more hospitalizations per year, than patients who were not forced to skip their medications.
“Socioeconomic inequality in respiratory health in the U.S. from 1959 to 2018,” by Adam Gaffney, M.D., M.P.H.; David U. Himmelstein, M.D.; David C. Christiani, M.D., M.S., M.P.H.; Steffie Woolhandler, M.D., M.P.H.; JAMA Internal Medicine, May 28, 2021. This new study suggests that poor lung health and higher rates of respiratory problems may have left lower-income Americans susceptible to the pneumonia often caused by the coronavirus. From 1959 to 2018, socioeconomic disparities in respiratory symptoms, lung disease prevalence, and pulmonary function mostly persisted — and in some instances appeared to widen — despite improvements in air quality and tobacco use, suggesting that the benefits of these improvements have not been equitably enjoyed. Researchers conclude that social class may function as an independent determinant of lung health.
“Racial and ethnic disparities in outpatient visit rates across 29 specialties,” Christopher Cai, M.D.; Adam Gaffney, M.D., M.P.H.; Alecia McGregor, Ph.D.; Steffie Woolhandler, M.D., M.P.H.; David U. Himmelstein, M.D.; Danny McCormick, M.D., M.P.H.; Samuel Dickman, M.D. JAMA Internal Medicine, July 19, 2021. Researchers found that people of color are underrepresented in the outpatient practices of most specialist physicians, including surgical specialists such as orthopedists and medical subspecialists such as pulmonary (lung) specialists. Disparities persisted even after accounting for patients’ insurance, income, education, and health status. For example, Black patients’ visit rates to orthopedic surgeons, urologists, pulmonologists, and cardiologists were 59%, 62%, 63%, and 81% those of white individuals, respectively. Notably, nephrologists — who care for patients with end-stage kidney disease, almost all of whom are covered by Medicare, and many of whom are people of color — provided significantly more care to minority groups than to whites. In contrast, primary care physicians saw patients of color and white patients at roughly equal rates.
“States’ performance in reducing uninsurance among Black, Hispanic, and low-income Americans following implementation of the Affordable Care Act,” Gregory Lines, Kira Mengistu, Megan Rose, Carr LaPorte, Deborah Lee, Lynn Anderson, Daniel Novinson, Erica Dwyer, Sonja Grigg, Hugo Torres, Gaurab Basu, and Danny McCormick, Health Equity, July 21, 2021. Gains in health insurance coverage under the Affordable Care Act (ACA) were small for Black, Hispanic and low-income Americans in many states. This study found dramatic variation in states’ performance in expanding insurance coverage to these populations that have historically had low coverage rates. While the best performing states were able to reduce rates of uninsurance among Black, Hispanic and low-income adults by approximately 60%, the worst performing states reduced uninsurance by less than 10%, a six-fold difference. The study also found that, two years after ACA implementation, in six states one quarter of Black adults remained without insurance coverage; in 20 states, one quarter of low-income adults continued to lack coverage; in 13 states, over 40% of Hispanic adults lacked coverage.
“Changes in racial and ethnic disparities in access to care and health among U.S. adults at age 65 years,” Jacob Wallace, Ph.D.; Karen Jiang, B.A.; Paul Goldsmith-Pinkham, Ph.D.; Zirui Song, M.D., Ph.D.; JAMA Internal Medicine, July 26, 2021. Immediately after age 65 years — the age at which all adults are eligible for Medicare coverage — disparities between white and Black adults and between white and Hispanic adults sharply decrease. After age 65, there were marked reductions in the share of the population that was uninsured, without a usual source of care, unable to see a physician in the past year owing to cost, and in poor self-reported health.
“Promise vs. Practice: The actual financial performance of Accountable Care Organizations,” James G. Khan, M.D., M.P.H. and Kip Sullivan, J.D., Journal of General Internal Medicine, August 13, 2021. The authors collect and compare financial performance data from all four CMS Accountable Care Organization (ACO) programs from 2005 to 2018, examining net CMS cost (gross savings in medical billings minus “bonus” payments to ACOs). They found that overall, ACO programs roughly broke even — from the CMS perspective. That is, when bonuses CMS paid to ACOs are subtracted from gross savings, the programs lost money or saved no more than a few tenths of a percent.
“We can heal from hate crimes by practicing solidarity,” by Christopher Cai, M.D., JAMA Network, June 21, 2021. Former SNaHP leader Dr. Chris Cai reflects on the experience of being a young Asian-American physician. He suggests that Asian- American physicians can heal from recent hate crimes by advocating for policies that dismantle structural racism in medicine and broader society.
“How would Medicare for All affect physician revenue?” by Christopher Cai, M.D., Journal of General Internal Medicine, July 8, 2021. Dr. Cai explains that the available evidence suggests physicians would prosper under single-payer reform. By supporting Medicare for All, physicians — and organized medicine — can both advocate for physicians’ self-interest while advancing legislation that would be enormously beneficial to patients.
“Financial profit in medicine: A position paper from the American College of Physicians,” Ryan Crowley, B.S.J.; Omar Atiq, M.D.; David Hilden, M.D.; Annal of Internal Medicine, September 7, 2021. In this position paper following the group’s endorsement of single-payer reforms, the ACP explains that profit motive in medicine may contribute to a bloated, complex, and fragmented health care system. “In recent years, we have seen health care become increasingly business-oriented with more for-profit entities and private equity investments,” said Thomas G. Cooney, M.D., chair of ACP’s Board of Regents. “We need to be sure that profits never become more important than patient care in the practice of medicine.”
In California, four physician members met with state Senator Monique Limon in July, sharing stories about patients who could have been helped by a single-payer plan. Sen. Limon is a member of the senate health committee. PNHP-CA members participated in several actions this summer, including a rally at the state capitol in support of the Healthy California Now bill, a rally demanding Gov. Newsom lead the way on single payer, a delegation to the national March for Medicare for All in Los Angeles, and an event celebrating Medicare’s birthday and demanding Medicare for All. To get involved in California, contact Dr. Kathleen Healey at khealey.ent@gmail.com.

Many Illinois members have been giving interviews and speeches about Medicare for All. Shannon Rotolo, PharmD, a leader in the Illinois Single-Payer Coalition and founder of Pharmacists for Single Payer, discussed pharmacy issues on a Healthcare-NOW! podcast, and was quoted in the Journal of the American Pharmacists Association. Co-president Dr. Pam Gronemeyer spoke about Medicare for all on a podcast with Chicago journalist Ben Joravsky, and at the Chicago March for Medicare for All. Dr. Anne Scheetz spoke to the LaSalle Democratic Central Committee and on a panel sponsored by Chicago Jobs with Justice and Illinois Single Payer Coalition. The chapter also participated in several other events this summer, including a Chicago vigil for global vaccine access; a rally for Medicare for All at the office of Rep. Raja Krishnamoorthi; and a campaign by the Chicago-based Jesse Brown VA Medical Center’s Clinical Committee for Black Lives, urging the VA to discontinue the use of race-based algorithms in kidney function calculations. To get involved in Illinois, contact Dr. Anne Scheetz at annescheetz@gmail.com.
In Kentucky, Kentuckians for Single Payer Health Care found that many seniors who switch back to Traditional Medicare from a commercial Medicare Advantage plan are not protected from being denied a Medigap plan, or charged more, because of pre-existing conditions such as age, health status, claims experience, or medical condition. Dr. Eugene Shively proposed and successfully passed a resolution at the August meeting of the Kentucky Medical Association calling on the state legislature to end Medigap discrimination. In addition to their work on Medigap plans, chapter members helped to organize the Louisville March for Medicare for All in July, earning local media coverage. To get involved in Kentucky, contact Kay Tillow at nursenpo@aol.com.
Maine’s legislature passed the Maine Health Care Act, authorizing the state to request waivers from the federal government to implement a state universal health program, and requiring that 60 days after the implementation of such waivers, the governor appoint a board to design the state plan. The bill is a result of hundreds of volunteer hours, including testimony, meetings, and the production of educational materials for legislators. PNHP members also participated in the March for Medicare for All in Portland, earning media coverage from one TV station and two of Maine’s largest newspapers. PNHP’ers in Maine are also gathering signatures for a ballot initiative directing the state legislature to establish a universal health care system in the state. To get involved in Maine, contact Dr. Henk Goorhuis at henk@maineallcare.org.

PNHP-Minnesota hosted a workshop in July on passing local government resolutions in support of Medicare for All. Participants heard from health care activists around the state working on active resolution campaigns, as well as those looking to start new campaigns. To get involved in Minnesota, contact pnhpminnesota@gmail.com.
PNHP’s New Jersey chapter hosted a planning session in June to discuss organizing strategies to win national Medicare for All. Speakers included U.S. Rep. Bonnie Watson Coleman, who is vice-chair of the Congressional Progressive Caucus. To get involved in New Jersey, contact Dr. William Thar at wethar@gmail.com.

In New York, PNHP’s NY-Metro chapter is continuing to focus on organizing around the New York Health Act, which was introduced this year with majority support in both the Assembly and the Senate. Organizing tactics included social media campaigns, as well as a series of in-person rallies outside of the offices of targeted legislators, along with a large rally and “die-in” civil disobedience outside of the state capitol building. This spring and summer, the chapter also focused on fighting a move by New York City to shift its public union retirees over to a Medicare Advantage plan. PNHP-NY Metro and the NY Statewide Senior Action Council worked with concerned union members, providing them with tools to coordinate the larger group of retirees interested in the fight, and by hosting two informational forums about the proposed change and how it would affect retirees. To get involved in New York, contact NY Metro Executive Director Bob Lederer at info@pnhpnymetro.org.
In North Carolina, board members of Health Care Justice NC — PNHP’s chapter in Charlotte — led several presentations on the topic of the pandemic, health inequities, and Medicare for All to the Atrium Health System pediatric residents, the staff of the Charlotte Center for Legal Advocacy, and to Duke University’s African-American Covid Taskforce Meeting on July 20. After a vote by the board, chapter members have been contacting their elected representatives to advocate for improvements to traditional Medicare, including lowering eligibility age to 60, coverage for dental, vision, and hearing care, a cap on out-of-pocket expenses, and allowing Medicare to negotiate drug prices. To get involved in Health Care Justice NC, contact Dr. Jessica Schorr Saxe at jessica.schorr.saxe@gmail.com.

Members of Health Care for All Western North Carolina (HCFA-WNC) in Asheville presented a Medicare for All resolution to the Asheville City Council and the Buncombe County Commission. Members also picketed in solidarity with NNU nurses Mission Hospital, who were organizing for a new contract. HCFA-WNC joined with six other organizations to plan and sponsor a March for Medicare for All which attracted more than 100 marchers. To get involved in HCFA-WNC, contact Terry Hash at theresamhash@gmail.com.

Health Care for All-NC co-hosted a teach-in with the Freelance Solidarity Project, a division of the National Writers Union, about why Medicare for All matters to all freelance workers, and how they can support it. Panelists included Rhiannon Duryea, the national coordinator for the Labor Campaign for Single Payer; Natalie Shure, a writer for The New Republic; and Dominic Harris, president of UE Local 150 in Charlotte and chair of UE 150’s Medicare for All campaign. PNHP is working with the writers’ union leadership to draft a resolution in support of Medicare for All. To get involved in Health Care for All NC, contact Jonathan Michels at jonscottmichels@gmail.com.

Members of PNHP Oregon helped to organize a March for Medicare for All rally in July. The chapter has also joined PNHP’s Medical Society Resolutions Campaign, with the goal of passing resolutions in local chapters of internal medicine and pediatrics specialty societies. Dr. Paul Gormann is helping to organize a new chapter of Students for a National Health Program at Oregon Health and Science University. To get involved in Oregon, contact Dr. Peter Mahr at peter.n.mahr@gmail.com.

In Pennsylvania, members of PNHP’s Philadelphia chapter met with the chief of staff of Rep. Dwight Evans to learn why he is hesitant to support H.R. 1976, even though he endorsed previous single-payer bills. The chapter will use his response to strategize next steps in their campaign to win support from all members of the area’s Congressional delegation. To get involved in Philadelphia, contact Dr. Walter Tsou at walter.tsou@verizon.net.
PNHP’s Western Pennsylvania chapter continues to ally with the local labor movement, and recently visited striking members of the United Steelworkers to build solidarity. The chapter also sponsored the Pittsburgh March for Medicare for All in July, which attracted about 100 single-payer activists. To get involved in Western PA, contact Dr. Judy Albert at jalbertpgh@gmail.com.
In Tennessee, PNHP’s State of Franklin chapter (which includes easternmost Tennessee and southwest Virginia) holds monthly Zoom meetings focused on developing single-payer messaging that will appeal to more conservative friends, family and neighbors. In the past year, guest speakers included PNHP past president Dr. Carol Paris and former insurance executive Wendell Potter. Members have also published multiple op-eds and letters to editors in local media, and joined in coalition with other non-physician groups fighting for Medicare for All. To get involved in Tennessee, contact Dr. Bob Funke at r_funke@charter.net or Dr. Robin Feierabend at robin@firerobin.net.
PNHP chapters in Vermont and New Hampshire completed their sixth annual summer internship program in July, with 14 rising second-year medical students. Because this year’s internship was online, students could hear from speakers across the country and globe. Topics included the market failures of health policy, the history of the U.S. health care system, the business model of the private insurance industry, and the history and potential of the Medicare, Medicaid and other public programs. The internship also included sessions on advocacy, such as organizing physicians and chapters, educating medical students, utilizing traditional and social media, and messages for legislators and the general public. To get involved in Vermont, contact Dr. Betty Keller at bjkellermd@gmail.com.

In Washington state, PNHP members held a session on the pros and cons of incremental change in the struggle for single payer. The chapter helped organize the March for Medicare for All in Seattle, and facilitated an opening greeting from Medicare for All lead sponsor Rep. Pramila Jayapal. Members hung Medicare for All banners over Seattle freeways on several occasions. To get involved in Washington, contact Dr. McLanahan at mcltan@comcast.net.
PNHP welcomes its first chapter in West Virginia, which formed in January 2021. The new chapter has been busy crafting by-laws, creating a 12-member board, and launching the first SNaHP chapter in the state. The chapter’s first project is a “55 strong” organizing campaign to recruit a PNHP member in each of the 55 counties of the mainly rural state. They are also actively recruiting in the West Virginia State Medical Association and working on Medicare for All municipal resolutions. To get involved in West Virginia, contact Dr. Daniel Doyle at doyledan348@gmail.com.
“Demonstrators push for ‘Medicare for All’ at west Toledo rally,” WTOL Ch. 11 News, May 22, 2021 [Ohio Chapter]
“Demonstrators in Louisville join nationwide Medicare for All March,” Spectrum News 1 (Louisville), July 24, 2021 [Kay Tillow and Dr. Garrett Adams]
“Thousands march to demand Medicare for All,” Free Speech TV/Rising up with Sonali, July 28, 2021 [Dr. Paul Song]
“Un-vaxxed tax: Should the unvaccinated have to pay more for health insurance?” MSNBC: All in with Chris Hayes, August 12, 2021 [Dr. Adam Gaffney]